Filter News
Area of Research



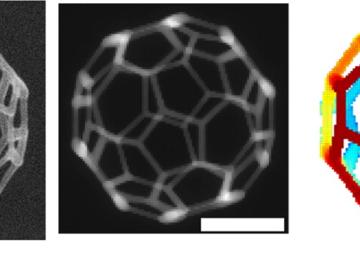

Scientists can now detect magnetic behavior at the atomic level with a new electron microscopy technique developed by a team from the Department of Energy’s Oak Ridge National Laboratory and Uppsala University, Sweden.

In a rechargeable battery, the electrolyte transports lithium ions from the negative to the positive electrode during discharging. The path of ionic flow reverses during recharging.
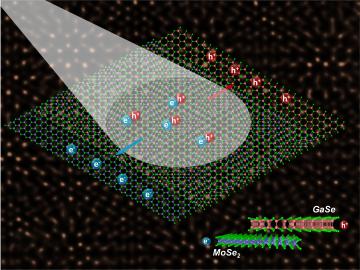
Epitaxy, or growing crystalline film layers that are templated by a crystalline substrate, is a mainstay of manufacturing transistors and semiconductors.
Researchers at the Department of Energy’s Oak Ridge National Laboratory have found a potential path to further improve solar cell efficiency by understanding the competition among halogen atoms during the synthesis of sunlight-absorbing crystals.