Filter News
Area of Research
- (-) Neutron Science (20)
- Biological Systems (1)
- Biology and Environment (23)
- Clean Energy (14)
- Computational Biology (2)
- Computational Engineering (1)
- Energy Frontier Research Centers (1)
- Fusion and Fission (2)
- Isotopes (5)
- Materials (46)
- Materials for Computing (8)
- National Security (3)
- Nuclear Science and Technology (2)
- Quantum information Science (1)
- Supercomputing (28)
News Topics
- (-) Biomedical (11)
- (-) Nanotechnology (10)
- 3-D Printing/Advanced Manufacturing (6)
- Advanced Reactors (1)
- Artificial Intelligence (6)
- Big Data (2)
- Bioenergy (6)
- Biology (5)
- Biotechnology (1)
- Chemical Sciences (2)
- Clean Water (2)
- Climate Change (1)
- Composites (1)
- Computer Science (13)
- Coronavirus (8)
- Cybersecurity (1)
- Decarbonization (2)
- Energy Storage (6)
- Environment (8)
- Fossil Energy (1)
- Frontier (1)
- Fusion (1)
- High-Performance Computing (2)
- Machine Learning (3)
- Materials (14)
- Materials Science (23)
- Mathematics (1)
- Microscopy (3)
- National Security (2)
- Neutron Science (99)
- Nuclear Energy (3)
- Physics (9)
- Polymers (1)
- Quantum Computing (1)
- Quantum Science (7)
- Security (2)
- Space Exploration (3)
- Summit (6)
- Sustainable Energy (2)
- Transportation (5)
Media Contacts
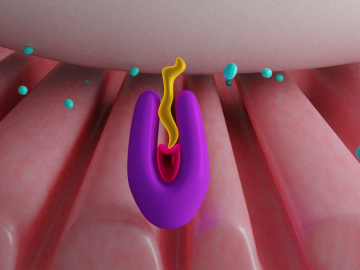
Pick your poison. It can be deadly for good reasons such as protecting crops from harmful insects or fighting parasite infection as medicine — or for evil as a weapon for bioterrorism. Or, in extremely diluted amounts, it can be used to enhance beauty.

Five researchers at the Department of Energy’s Oak Ridge National Laboratory have been named ORNL Corporate Fellows in recognition of significant career accomplishments and continued leadership in their scientific fields.
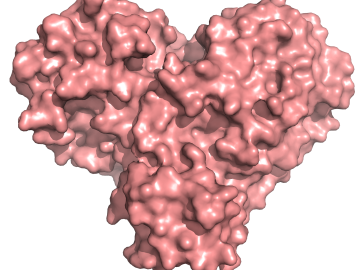
A team of researchers has performed the first room-temperature X-ray measurements on the SARS-CoV-2 main protease — the enzyme that enables the virus to reproduce.
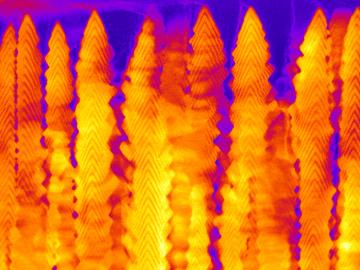
A team led by the Department of Energy’s Oak Ridge National Laboratory synthesized a tiny structure with high surface area and discovered how its unique architecture drives ions across interfaces to transport energy or information.

Matthew R. Ryder, a researcher at the Department of Energy’s Oak Ridge National Laboratory, has been named the 2020 Foresight Fellow in Molecular-Scale Engineering.
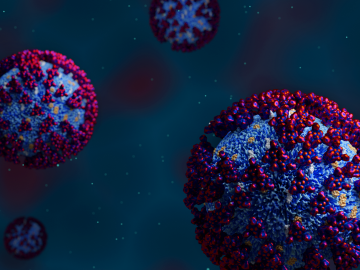
In the race to identify solutions to the COVID-19 pandemic, researchers at the Department of Energy’s Oak Ridge National Laboratory are joining the fight by applying expertise in computational science, advanced manufacturing, data science and neutron science.

Biological membranes, such as the “walls” of most types of living cells, primarily consist of a double layer of lipids, or “lipid bilayer,” that forms the structure, and a variety of embedded and attached proteins with highly specialized functions, including proteins that rapidly and selectively transport ions and molecules in and out of the cell.
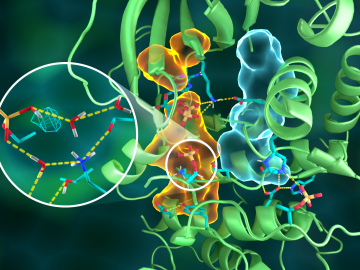
OAK RIDGE, Tenn., March 20, 2019—Direct observations of the structure and catalytic mechanism of a prototypical kinase enzyme—protein kinase A or PKA—will provide researchers and drug developers with significantly enhanced abilities to understand and treat fatal diseases and neurological disorders such as cancer, diabetes, and cystic fibrosis.
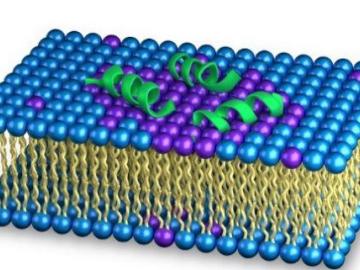
As the rise of antibiotic-resistant bacteria known as superbugs threatens public health, Oak Ridge National Laboratory’s Shuo Qian and Veerendra Sharma from the Bhaba Atomic Research Centre in India are using neutron scattering to study how an antibacterial peptide interacts with and fights harmful bacteria.
![2018-P07635 BL-6 user - Univ of Guelph-6004R_sm[2].jpg 2018-P07635 BL-6 user - Univ of Guelph-6004R_sm[2].jpg](/sites/default/files/styles/list_page_thumbnail/public/2018-P07635%20BL-6%20user%20-%20Univ%20of%20Guelph-6004R_sm%5B2%5D.jpg?itok=DUdZNt_q)
A team of scientists, led by University of Guelph professor John Dutcher, are using neutrons at ORNL’s Spallation Neutron Source to unlock the secrets of natural nanoparticles that could be used to improve medicines.




