
In a new twist to waste-to-fuel technology, ORNL scientists have developed an electrochemical process that uses tiny spikes of carbon and copper to turn carbon dioxide, a greenhouse gas, into ethanol.

In a new twist to waste-to-fuel technology, ORNL scientists have developed an electrochemical process that uses tiny spikes of carbon and copper to turn carbon dioxide, a greenhouse gas, into ethanol.


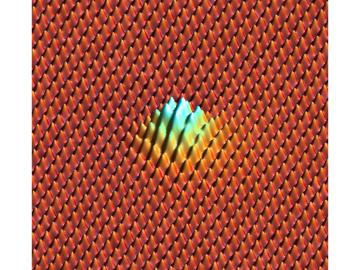
Researchers at Oak Ridge National Laboratory found a simpler way to measure adhesion between graphene sheets, compared to a sophisticated method used in a 2015 study: They measured how much graphene deflects when neon atoms poke it from below to create

Ceramic matrix composite (CMC) materials are made of coated ceramic fibers surrounded by a ceramic matrix. They are tough, lightweight and capable of withstanding temperatures 300–400 degrees F hotter than metal alloys can endure.

