An Oak Ridge National Laboratory–led team discovered that vanadium dioxide in a crystalline thin film makes an outstanding electrode for lithium-ion batteries.



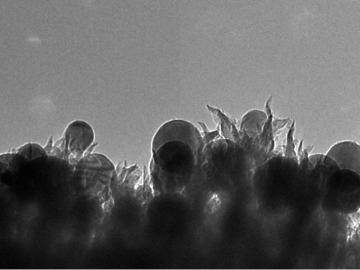
In a new twist to waste-to-fuel technology, ORNL scientists have developed an electrochemical process that uses tiny spikes of carbon and copper to turn carbon dioxide, a greenhouse gas, into ethanol.

Researchers have long sought electrically conductive materials for economical energy-storage devices. Two-dimensional (2D) ceramics called MXenes are contenders.

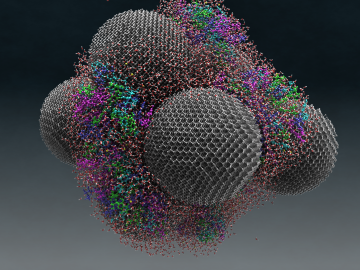

In a new twist to waste-to-fuel technology, ORNL scientists have developed an electrochemical process that uses tiny spikes of carbon and copper to turn carbon dioxide, a greenhouse gas, into ethanol.