Filter News
Area of Research
News Type

Scientists at Oak Ridge National Laboratory have conducted a series of breakthrough experimental and computational studies that cast doubt on a 40-year-old theory describing how polymers in plastic materials behave during processing.


A scientific team led by the Department of Energy’s Oak Ridge National Laboratory has found a new way to take the local temperature of a material from an area about a billionth of a meter wide, or approximately 100,000 times thinner than a human hair.
A new method to produce large, monolayer single-crystal-like graphene films more than a foot long relies on harnessing a “survival of the fittest” competition among crystals.



Material surfaces and interfaces may appear flat and void of texture to the naked eye, but a view from the nanoscale reveals an intricate tapestry of atomic patterns that control the reactions between the material and its environment.
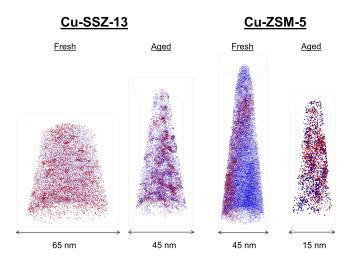
Diesel vehicles today emit far fewer pollutants than older vehicles, thanks to a zeolite (hydrous silicate) catalytic converter that was invented around 10 years ago to reduce pollutants that cause the formation of acid rain and smog.

Five researchers at the Department of Energy’s Oak Ridge National Laboratory have been elected fellows of the American Association for the Advancement of Science (AAAS). AAAS, the world’s largest multidisciplinary scientific society

