Filter News
Area of Research
- (-) Isotopes (2)
- (-) Neutron Science (10)
- Advanced Manufacturing (14)
- Biology and Environment (34)
- Building Technologies (1)
- Clean Energy (77)
- Climate and Environmental Systems (3)
- Computational Biology (1)
- Computational Engineering (2)
- Computer Science (7)
- Electricity and Smart Grid (2)
- Functional Materials for Energy (2)
- Fusion and Fission (8)
- Fusion Energy (6)
- Materials (50)
- Materials Characterization (2)
- Materials for Computing (9)
- Materials Under Extremes (1)
- Mathematics (1)
- National Security (4)
- Nuclear Science and Technology (7)
- Quantum information Science (4)
- Sensors and Controls (1)
- Supercomputing (21)
News Type
News Topics
- (-) Biomedical (3)
- (-) Environment (3)
- (-) Fusion (1)
- (-) Materials (6)
- (-) Quantum Science (2)
- Advanced Reactors (1)
- Artificial Intelligence (1)
- Bioenergy (1)
- Biology (2)
- Chemical Sciences (1)
- Climate Change (2)
- Computer Science (2)
- Cybersecurity (1)
- Energy Storage (3)
- Irradiation (1)
- Isotopes (6)
- Materials Science (8)
- Microscopy (1)
- Nanotechnology (3)
- National Security (1)
- Neutron Science (32)
- Nuclear Energy (2)
- Physics (2)
- Security (1)
- Space Exploration (3)
- Transportation (1)
Media Contacts

Warming a crystal of the mineral fresnoite, ORNL scientists discovered that excitations called phasons carried heat three times farther and faster than phonons, the excitations that usually carry heat through a material.
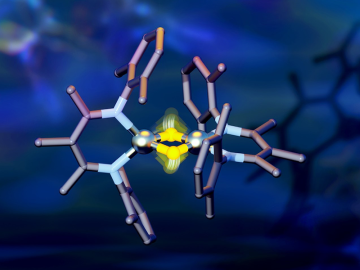
Researchers from Yale University and ORNL collaborated on neutron scattering experiments to study hydrogen atom locations and their effects on iron in a compound similar to those commonly used in industrial catalysts.

Three scientists from the Department of Energy’s Oak Ridge National Laboratory have been elected fellows of the American Association for the Advancement of Science, or AAAS.
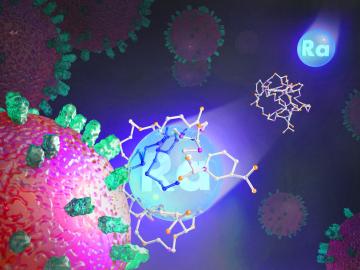
Researchers at ORNL explored radium’s chemistry to advance cancer treatments using ionizing radiation.


The Department of Energy’s Office of Science has selected five Oak Ridge National Laboratory scientists for Early Career Research Program awards.

The Department of Energy’s Office of Science has selected three Oak Ridge National Laboratory scientists for Early Career Research Program awards.

Matthew R. Ryder, a researcher at the Department of Energy’s Oak Ridge National Laboratory, has been named the 2020 Foresight Fellow in Molecular-Scale Engineering.
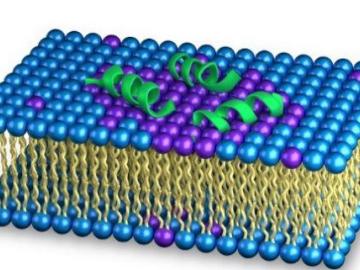
As the rise of antibiotic-resistant bacteria known as superbugs threatens public health, Oak Ridge National Laboratory’s Shuo Qian and Veerendra Sharma from the Bhaba Atomic Research Centre in India are using neutron scattering to study how an antibacterial peptide interacts with and fights harmful bacteria.
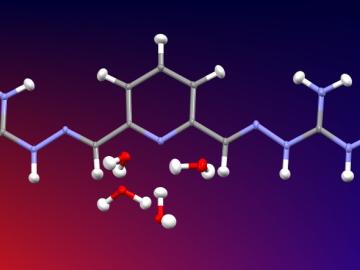
Researchers used neutron scattering at Oak Ridge National Laboratory’s Spallation Neutron Source to investigate the effectiveness of a novel crystallization method to capture carbon dioxide directly from the air.




