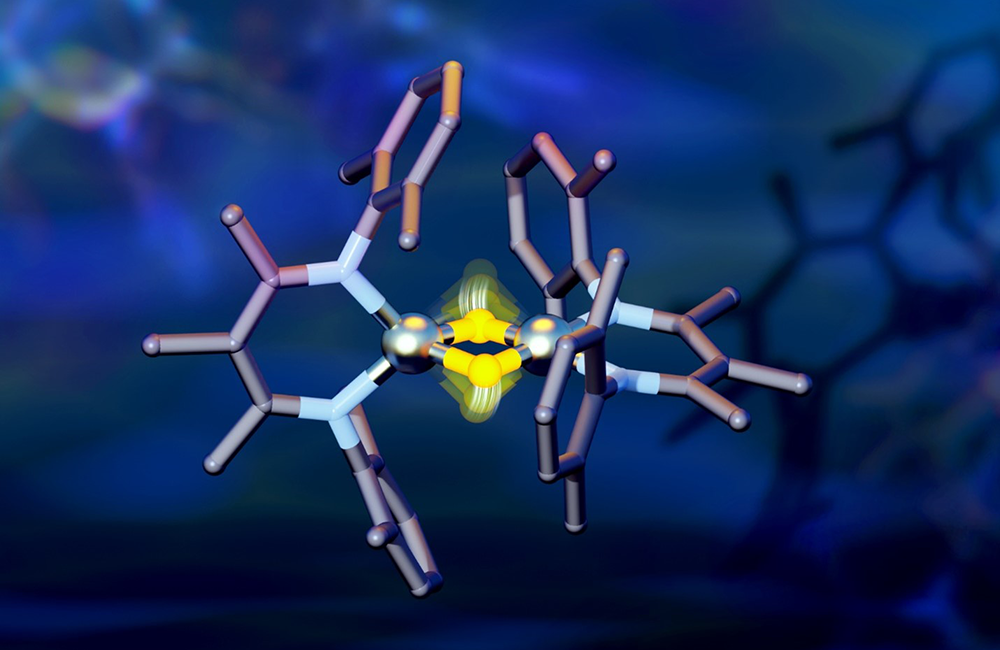
Even small movements of hydrogen, shown in yellow, were found to cause large energy shifts in the attached iron atoms, shown in silver, which could be of interest in creating novel chemical reactions. Credit: Jill Hemman/ORNL, U.S. Dept. of Energy
Researchers from Yale University and Oak Ridge National Laboratory collaborated on neutron scattering experiments to study hydrogen atom locations and their effects on iron in a compound similar to those commonly used in industrial catalysts.
In the study, the researchers discovered that hydrogen atoms move around the core of the molecule rapidly, causing the energy levels of electrons in the attached iron atoms to shift dramatically. The speed of their movement was controlled by slight changes in the surroundings of the molecule, showing surprising sensitivity.
“This suggests that hydrogen can be used to tune the dynamics and the electronic structure of iron, which could be harnessed to create or improve chemical reactions,” said Patrick Holland, professor of chemistry at Yale.
Neutron diffraction experiments at ORNL’s Spallation Neutron Source were used to locate the precise positions of the hydrogen atoms. The data will also be useful for novel catalyst designs.