Filter News
Area of Research
- (-) Isotope Development and Production (1)
- (-) Neutron Science (33)
- Advanced Manufacturing (5)
- Biological Systems (1)
- Biology and Environment (64)
- Biology and Soft Matter (1)
- Clean Energy (62)
- Climate and Environmental Systems (2)
- Computational Biology (2)
- Computational Engineering (2)
- Computer Science (1)
- Electricity and Smart Grid (1)
- Functional Materials for Energy (1)
- Fusion and Fission (26)
- Fusion Energy (13)
- Isotopes (8)
- Materials (102)
- Materials Characterization (1)
- Materials for Computing (18)
- Materials Under Extremes (1)
- Mathematics (1)
- National Security (18)
- Nuclear Science and Technology (17)
- Sensors and Controls (1)
- Supercomputing (55)
- Transportation Systems (1)
News Topics
- (-) Biomedical (11)
- (-) Climate Change (1)
- (-) Fusion (1)
- (-) Materials Science (24)
- (-) Polymers (1)
- (-) Security (2)
- 3-D Printing/Advanced Manufacturing (6)
- Advanced Reactors (1)
- Artificial Intelligence (6)
- Big Data (2)
- Bioenergy (6)
- Biology (5)
- Biotechnology (1)
- Chemical Sciences (2)
- Clean Water (2)
- Composites (1)
- Computer Science (13)
- Coronavirus (8)
- Cybersecurity (1)
- Decarbonization (2)
- Energy Storage (6)
- Environment (8)
- Fossil Energy (1)
- Frontier (1)
- High-Performance Computing (2)
- Irradiation (1)
- Machine Learning (3)
- Materials (14)
- Mathematics (1)
- Microscopy (3)
- Nanotechnology (10)
- National Security (2)
- Neutron Science (99)
- Nuclear Energy (4)
- Physics (9)
- Quantum Computing (1)
- Quantum Science (7)
- Space Exploration (4)
- Summit (6)
- Sustainable Energy (2)
- Transportation (5)
Media Contacts

Five researchers at the Department of Energy’s Oak Ridge National Laboratory have been named ORNL Corporate Fellows in recognition of significant career accomplishments and continued leadership in their scientific fields.
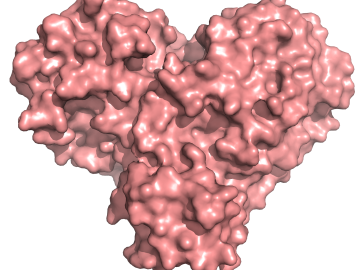
A team of researchers has performed the first room-temperature X-ray measurements on the SARS-CoV-2 main protease — the enzyme that enables the virus to reproduce.
A team led by the Department of Energy’s Oak Ridge National Laboratory synthesized a tiny structure with high surface area and discovered how its unique architecture drives ions across interfaces to transport energy or information.

Research by an international team led by Duke University and the Department of Energy’s Oak Ridge National Laboratory scientists could speed the way to safer rechargeable batteries for consumer electronics such as laptops and cellphones.
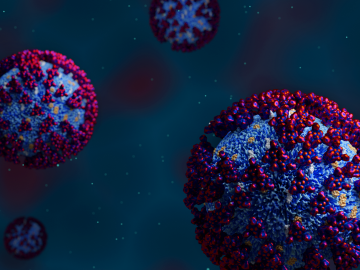
In the race to identify solutions to the COVID-19 pandemic, researchers at the Department of Energy’s Oak Ridge National Laboratory are joining the fight by applying expertise in computational science, advanced manufacturing, data science and neutron science.

Biological membranes, such as the “walls” of most types of living cells, primarily consist of a double layer of lipids, or “lipid bilayer,” that forms the structure, and a variety of embedded and attached proteins with highly specialized functions, including proteins that rapidly and selectively transport ions and molecules in and out of the cell.
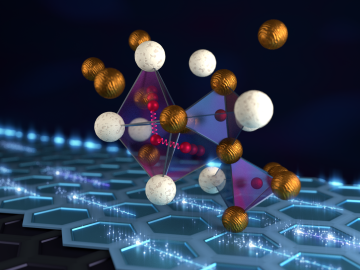
An international team of researchers has discovered the hydrogen atoms in a metal hydride material are much more tightly spaced than had been predicted for decades — a feature that could possibly facilitate superconductivity at or near room temperature and pressure.

Scientists at the U.S. Department of Energy’s Brookhaven National Laboratory have new experimental evidence and a predictive theory that solves a long-standing materials science mystery: why certain crystalline materials shrink when heated.
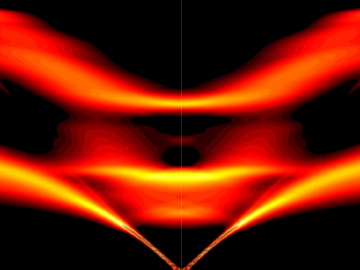
Scientists have discovered a way to alter heat transport in thermoelectric materials, a finding that may ultimately improve energy efficiency as the materials
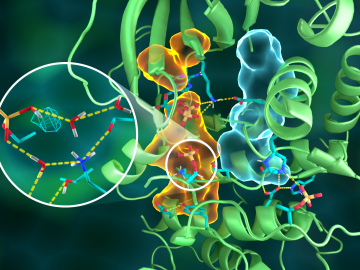
OAK RIDGE, Tenn., March 20, 2019—Direct observations of the structure and catalytic mechanism of a prototypical kinase enzyme—protein kinase A or PKA—will provide researchers and drug developers with significantly enhanced abilities to understand and treat fatal diseases and neurological disorders such as cancer, diabetes, and cystic fibrosis.




