Filter News
Area of Research
- (-) Materials for Computing (10)
- (-) Neutron Science (23)
- (-) Quantum information Science (3)
- Advanced Manufacturing (2)
- Biology and Environment (96)
- Biology and Soft Matter (1)
- Clean Energy (73)
- Climate and Environmental Systems (5)
- Computational Engineering (1)
- Computer Science (2)
- Electricity and Smart Grid (1)
- Energy Frontier Research Centers (1)
- Functional Materials for Energy (1)
- Fusion and Fission (6)
- Fusion Energy (1)
- Isotope Development and Production (1)
- Isotopes (26)
- Materials (65)
- Mathematics (1)
- National Security (24)
- Nuclear Science and Technology (10)
- Supercomputing (63)
News Topics
- (-) Clean Water (2)
- (-) Cybersecurity (3)
- (-) Environment (9)
- (-) Frontier (1)
- (-) Isotopes (1)
- (-) Nanotechnology (18)
- (-) Space Exploration (3)
- 3-D Printing/Advanced Manufacturing (10)
- Advanced Reactors (1)
- Artificial Intelligence (6)
- Big Data (2)
- Bioenergy (8)
- Biology (6)
- Biomedical (13)
- Biotechnology (1)
- Chemical Sciences (6)
- Climate Change (2)
- Composites (2)
- Computer Science (24)
- Coronavirus (11)
- Decarbonization (3)
- Energy Storage (10)
- Fossil Energy (1)
- Fusion (1)
- Grid (1)
- High-Performance Computing (2)
- Machine Learning (3)
- Materials (24)
- Materials Science (35)
- Mathematics (1)
- Microscopy (9)
- National Security (3)
- Neutron Science (101)
- Nuclear Energy (3)
- Physics (10)
- Polymers (7)
- Quantum Computing (1)
- Quantum Science (18)
- Security (2)
- Simulation (1)
- Summit (6)
- Sustainable Energy (8)
- Transportation (9)
Media Contacts

Led by ORNL and the University of Tennessee, Knoxville, a study of a solar-energy material with a bright future revealed a way to slow phonons, the waves that transport heat.

Four research teams from the Department of Energy’s Oak Ridge National Laboratory and their technologies have received 2020 R&D 100 Awards.
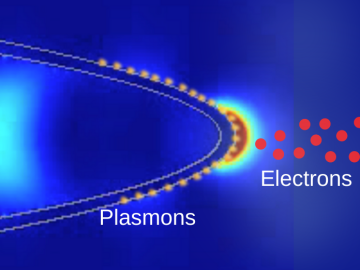
Scientists at ORNL and the University of Nebraska have developed an easier way to generate electrons for nanoscale imaging and sensing, providing a useful new tool for material science, bioimaging and fundamental quantum research.

Five researchers at the Department of Energy’s Oak Ridge National Laboratory have been named ORNL Corporate Fellows in recognition of significant career accomplishments and continued leadership in their scientific fields.

Scientists at ORNL used neutron scattering and supercomputing to better understand how an organic solvent and water work together to break down plant biomass, creating a pathway to significantly improve the production of renewable

The Department of Energy’s Office of Science has selected three Oak Ridge National Laboratory scientists for Early Career Research Program awards.
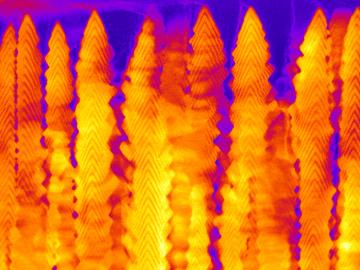
A team led by the Department of Energy’s Oak Ridge National Laboratory synthesized a tiny structure with high surface area and discovered how its unique architecture drives ions across interfaces to transport energy or information.

Matthew R. Ryder, a researcher at the Department of Energy’s Oak Ridge National Laboratory, has been named the 2020 Foresight Fellow in Molecular-Scale Engineering.
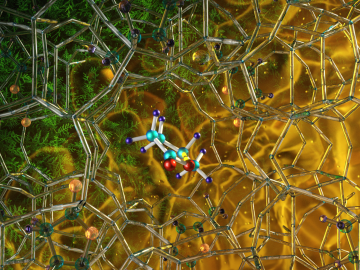
Illustration of the optimized zeolite catalyst, or NbAlS-1, which enables a highly efficient chemical reaction to create butene, a renewable source of energy, without expending high amounts of energy for the conversion. Credit: Jill Hemman, Oak Ridge National Laboratory/U.S. Dept. of Energy
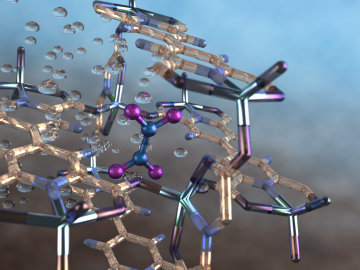
An international team of scientists, led by the University of Manchester, has developed a metal-organic framework, or MOF, material

