Filter News
Area of Research
News Topics
- (-) Cybersecurity (1)
- (-) Energy Storage (5)
- (-) Environment (3)
- (-) Machine Learning (1)
- (-) Microscopy (3)
- (-) Neutron Science (8)
- (-) Polymers (2)
- (-) Summit (1)
- 3-D Printing/Advanced Manufacturing (3)
- Artificial Intelligence (1)
- Bioenergy (4)
- Chemical Sciences (2)
- Climate Change (1)
- Composites (1)
- Computer Science (5)
- Critical Materials (2)
- Isotopes (2)
- Materials (1)
- Materials Science (22)
- Molten Salt (1)
- Nanotechnology (11)
- National Security (1)
- Physics (4)
- Quantum Science (6)
- Sustainable Energy (6)
- Transportation (2)
Media Contacts

Real-time measurements captured by researchers at ORNL provide missing insight into chemical separations to recover cobalt, a critical raw material used to make batteries and magnets for modern technologies.

Five researchers at the Department of Energy’s Oak Ridge National Laboratory have been named ORNL Corporate Fellows in recognition of significant career accomplishments and continued leadership in their scientific fields.
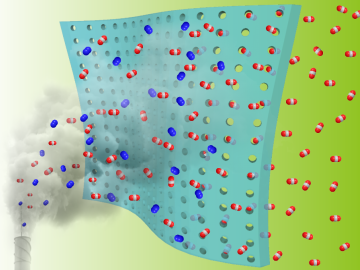
Researchers at the Department of Energy’s Oak Ridge National Laboratory and the University of Tennessee, Knoxville, are advancing gas membrane materials to expand practical technology options for reducing industrial carbon emissions.
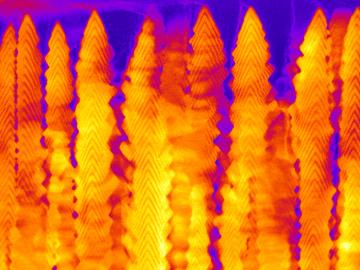
A team led by the Department of Energy’s Oak Ridge National Laboratory synthesized a tiny structure with high surface area and discovered how its unique architecture drives ions across interfaces to transport energy or information.

OAK RIDGE, Tenn., Feb. 27, 2020 — Researchers at Oak Ridge National Laboratory and the University of Tennessee achieved a rare look at the inner workings of polymer self-assembly at an oil-water interface to advance materials for neuromorphic computing and bio-inspired technologies.

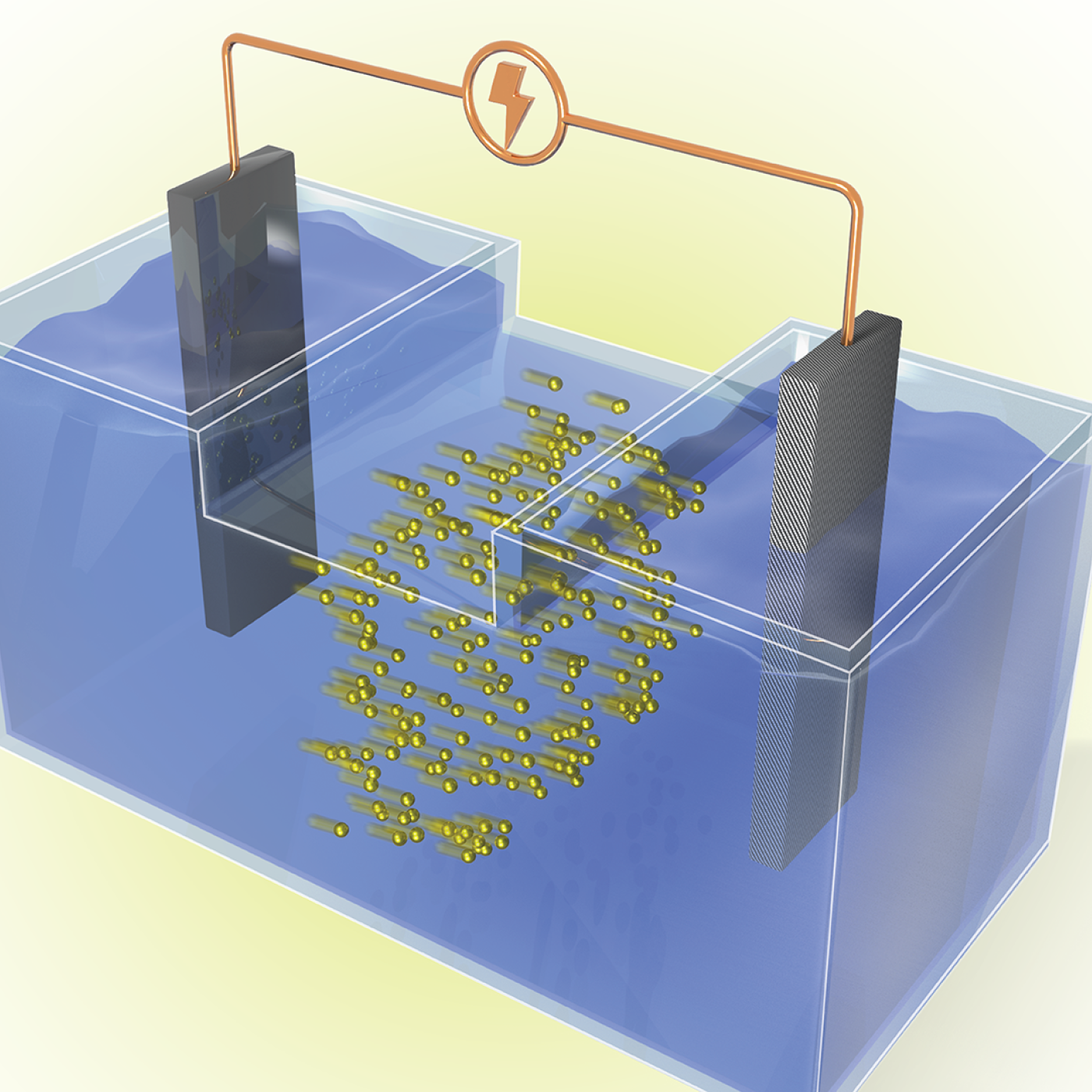
Energy storage startup SPARKZ Inc. has exclusively licensed five battery technologies from the Department of Energy’s Oak Ridge National Laboratory designed to eliminate cobalt metal in lithium-ion batteries. The advancement is aimed at accelerating the production of electric vehicles and energy storage solutions for the power grid.
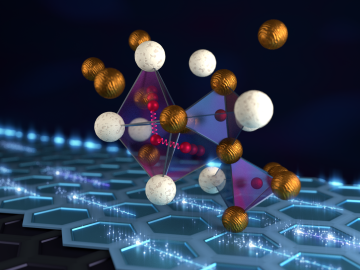
An international team of researchers has discovered the hydrogen atoms in a metal hydride material are much more tightly spaced than had been predicted for decades — a feature that could possibly facilitate superconductivity at or near room temperature and pressure.

Researchers at the Department of Energy’s Oak Ridge National Laboratory have received five 2019 R&D 100 Awards, increasing the lab’s total to 221 since the award’s inception in 1963.
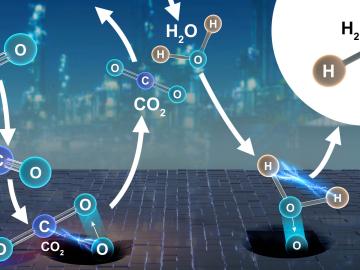
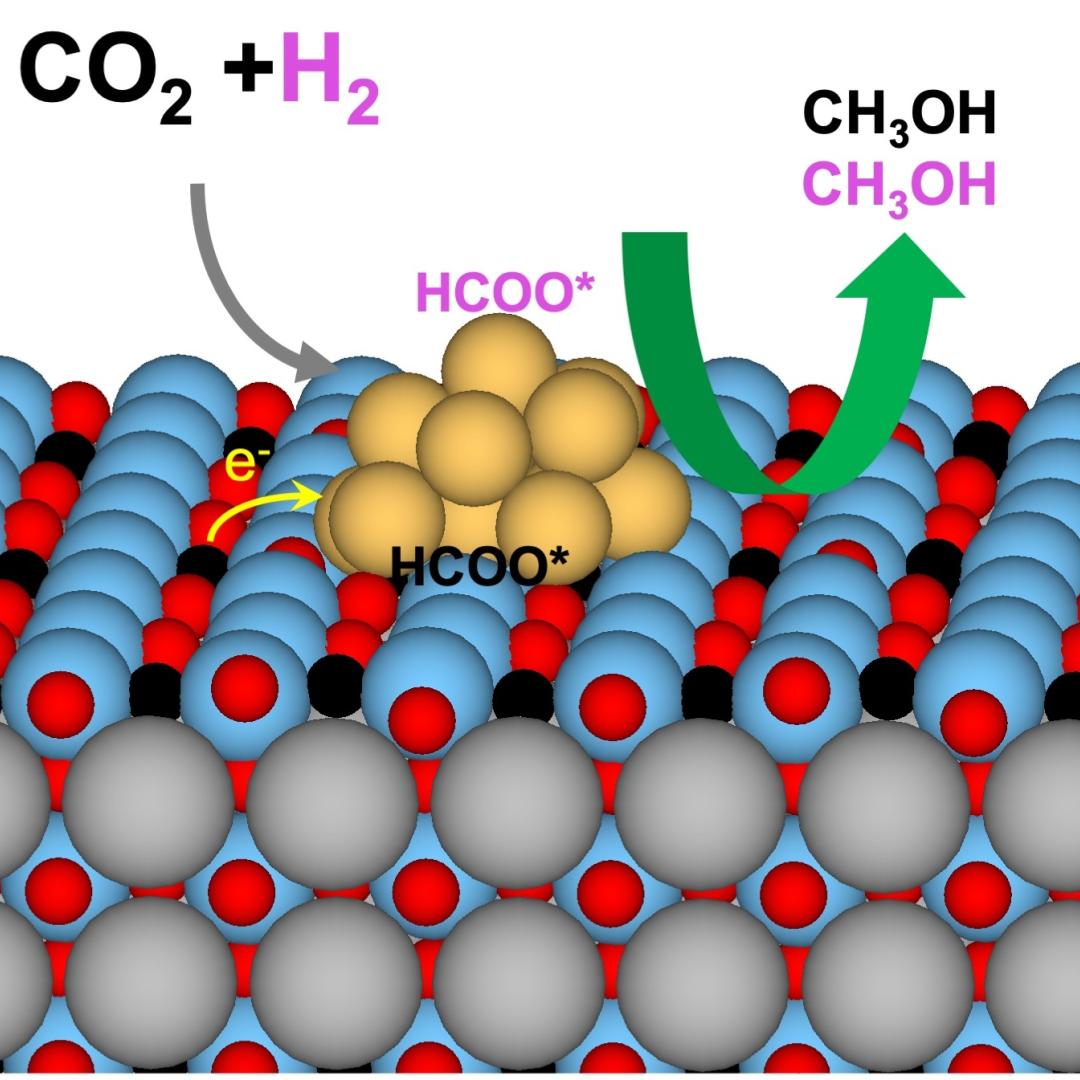
Collaborators at the Department of Energy’s Oak Ridge National Laboratory and U.S. universities used neutron scattering and other advanced characterization techniques to study how a prominent catalyst enables the “water-gas shift” reaction to purify and generate hydrogen at industrial scale.