Filter News
Area of Research
News Topics
- (-) Artificial Intelligence (1)
- (-) Biomedical (6)
- (-) Isotopes (6)
- (-) Machine Learning (1)
- (-) Microscopy (9)
- (-) Quantum Science (3)
- 3-D Printing/Advanced Manufacturing (7)
- Advanced Reactors (5)
- Bioenergy (4)
- Clean Water (2)
- Composites (3)
- Computer Science (6)
- Cybersecurity (1)
- Energy Storage (7)
- Environment (7)
- Fusion (4)
- Grid (1)
- Materials Science (26)
- Molten Salt (4)
- Nanotechnology (13)
- Neutron Science (24)
- Nuclear Energy (19)
- Physics (7)
- Polymers (5)
- Space Exploration (3)
- Sustainable Energy (5)
- Transportation (7)
Media Contacts

ORNL computer scientist Catherine Schuman returned to her alma mater, Harriman High School, to lead Hour of Code activities and talk to students about her job as a researcher.

Students often participate in internships and receive formal training in their chosen career fields during college, but some pursue professional development opportunities even earlier.
Scientists have discovered a way to alter heat transport in thermoelectric materials, a finding that may ultimately improve energy efficiency as the materials

A team led by scientists at the Department of Energy’s Oak Ridge National Laboratory explored how atomically thin two-dimensional (2D) crystals can grow over 3D objects and how the curvature of those objects can stretch and strain the

OAK RIDGE, Tenn., May 7, 2019—Energy Secretary Rick Perry, Congressman Chuck Fleischmann and lab officials today broke ground on a multipurpose research facility that will provide state-of-the-art laboratory space

OAK RIDGE, Tenn., March 22, 2019 – Karren Leslie More, a researcher at the Department of Energy’s Oak Ridge National Laboratory, has been elected fellow of the Microscopy Society of America (MSA) professional organization.
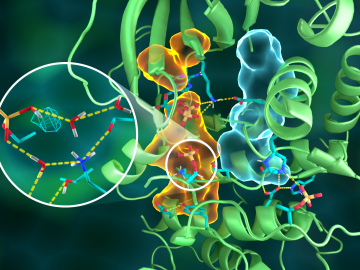
OAK RIDGE, Tenn., March 20, 2019—Direct observations of the structure and catalytic mechanism of a prototypical kinase enzyme—protein kinase A or PKA—will provide researchers and drug developers with significantly enhanced abilities to understand and treat fatal diseases and neurological disorders such as cancer, diabetes, and cystic fibrosis.
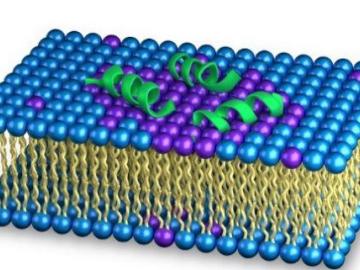
As the rise of antibiotic-resistant bacteria known as superbugs threatens public health, Oak Ridge National Laboratory’s Shuo Qian and Veerendra Sharma from the Bhaba Atomic Research Centre in India are using neutron scattering to study how an antibacterial peptide interacts with and fights harmful bacteria.

Vera Bocharova at the Department of Energy’s Oak Ridge National Laboratory investigates the structure and dynamics of soft materials—polymer nanocomposites, polymer electrolytes and biological macromolecules—to advance materials and technologies for energy, medicine and other applications.

OAK RIDGE, Tenn., Jan. 31, 2019—A new electron microscopy technique that detects the subtle changes in the weight of proteins at the nanoscale—while keeping the sample intact—could open a new pathway for deeper, more comprehensive studies of the basic building blocks of life.