Filter News
Area of Research
- (-) Isotopes (1)
- (-) Neutron Science (10)
- Advanced Manufacturing (1)
- Biology and Environment (19)
- Clean Energy (106)
- Computer Science (4)
- Electricity and Smart Grid (3)
- Energy Sciences (1)
- Functional Materials for Energy (2)
- Fusion and Fission (6)
- Materials (60)
- Materials for Computing (8)
- National Security (24)
- Nuclear Science and Technology (1)
- Quantum information Science (4)
- Sensors and Controls (1)
- Supercomputing (23)
News Topics
- (-) Cybersecurity (1)
- (-) Energy Storage (7)
- (-) Microscopy (3)
- 3-D Printing/Advanced Manufacturing (6)
- Advanced Reactors (1)
- Artificial Intelligence (6)
- Big Data (2)
- Bioenergy (6)
- Biology (5)
- Biomedical (16)
- Biotechnology (1)
- Chemical Sciences (2)
- Clean Water (2)
- Climate Change (2)
- Composites (1)
- Computer Science (13)
- Coronavirus (8)
- Decarbonization (2)
- Environment (9)
- Fossil Energy (1)
- Frontier (1)
- Fusion (1)
- High-Performance Computing (2)
- Irradiation (1)
- Isotopes (24)
- Machine Learning (3)
- Materials (18)
- Materials Science (24)
- Mathematics (1)
- Nanotechnology (10)
- National Security (3)
- Neutron Science (99)
- Nuclear Energy (6)
- Physics (9)
- Polymers (1)
- Quantum Computing (1)
- Quantum Science (7)
- Security (2)
- Space Exploration (6)
- Summit (6)
- Sustainable Energy (2)
- Transportation (5)
Media Contacts
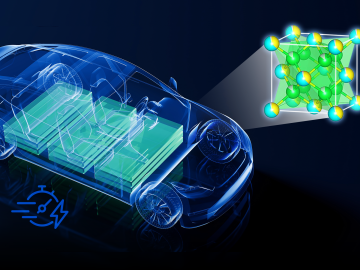
Currently, the biggest hurdle for electric vehicles, or EVs, is the development of advanced battery technology to extend driving range, safety and reliability.

Researchers at ORNL have developed a new method for producing a key component of lithium-ion batteries. The result is a more affordable battery from a faster, less wasteful process that uses less toxic material.
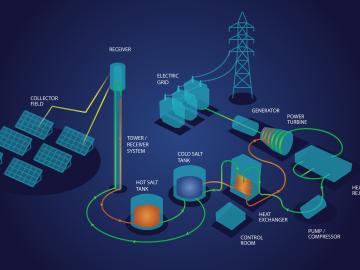
Oak Ridge National Laboratory scientists recently demonstrated a low-temperature, safe route to purifying molten chloride salts that minimizes their ability to corrode metals. This method could make the salts useful for storing energy generated from the sun’s heat.

Researchers at ORNL and the University of Tennessee, Knoxville, discovered a key material needed for fast-charging lithium-ion batteries. The commercially relevant approach opens a potential pathway to improve charging speeds for electric vehicles.

Three ORNL scientists have been elected fellows of the American Association for the Advancement of Science, or AAAS, the world’s largest general scientific society and publisher of the Science family of journals.

At the Department of Energy’s Oak Ridge National Laboratory, scientists use artificial intelligence, or AI, to accelerate the discovery and development of materials for energy and information technologies.

Five researchers at the Department of Energy’s Oak Ridge National Laboratory have been named ORNL Corporate Fellows in recognition of significant career accomplishments and continued leadership in their scientific fields.

Two of the researchers who share the Nobel Prize in Chemistry announced Wednesday—John B. Goodenough of the University of Texas at Austin and M. Stanley Whittingham of Binghamton University in New York—have research ties to ORNL.
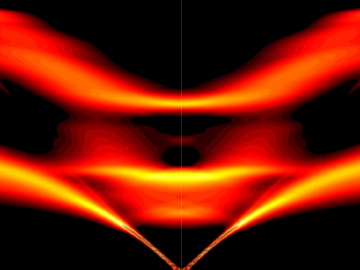
Scientists have discovered a way to alter heat transport in thermoelectric materials, a finding that may ultimately improve energy efficiency as the materials
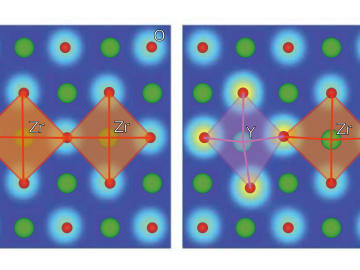
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.




