Filter News
Area of Research
- (-) Clean Energy (12)
- (-) Neutron Science (5)
- (-) Nuclear Science and Technology (2)
- Advanced Manufacturing (4)
- Biology and Environment (3)
- Computational Biology (1)
- Fusion and Fission (2)
- Fusion Energy (6)
- Isotopes (1)
- Materials (23)
- Materials for Computing (7)
- National Security (1)
- Supercomputing (7)
- Transportation Systems (1)
News Topics
- (-) Biomedical (4)
- (-) Fusion (1)
- (-) Materials Science (12)
- (-) Microscopy (3)
- (-) Simulation (1)
- 3-D Printing/Advanced Manufacturing (23)
- Advanced Reactors (4)
- Artificial Intelligence (2)
- Big Data (1)
- Bioenergy (3)
- Biology (2)
- Biotechnology (1)
- Buildings (13)
- Chemical Sciences (2)
- Clean Water (4)
- Climate Change (6)
- Composites (9)
- Computer Science (9)
- Coronavirus (2)
- Critical Materials (4)
- Decarbonization (4)
- Energy Storage (23)
- Environment (16)
- Grid (15)
- High-Performance Computing (1)
- Hydropower (2)
- Isotopes (1)
- Machine Learning (2)
- Materials (15)
- Mathematics (1)
- Mercury (1)
- Molten Salt (3)
- Nanotechnology (2)
- Net Zero (1)
- Neutron Science (24)
- Nuclear Energy (10)
- Physics (1)
- Polymers (5)
- Quantum Science (1)
- Space Exploration (4)
- Statistics (1)
- Sustainable Energy (27)
- Transportation (27)
Media Contacts

Warming a crystal of the mineral fresnoite, ORNL scientists discovered that excitations called phasons carried heat three times farther and faster than phonons, the excitations that usually carry heat through a material.

When aging vehicle batteries lack the juice to power your car anymore, they may still hold energy. Yet it’s tough to find new uses for lithium-ion batteries with different makers, ages and sizes. A solution is urgently needed because battery recycling options are scarce.

Scientists at Oak Ridge National Laboratory have developed a solvent that results in a more environmentally friendly process to recover valuable materials from used lithium-ion batteries, supports a stable domestic supply chain for new batteries

Oak Ridge National Laboratory researchers have demonstrated that a new class of superalloys made of cobalt and nickel remains crack-free and defect-resistant in extreme heat, making them conducive for use in metal-based 3D printing applications.

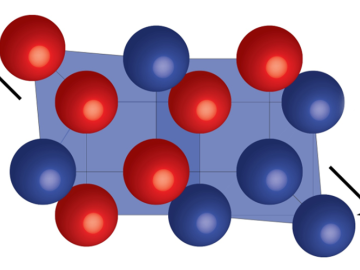
Pauling’s Rules is the standard model used to describe atomic arrangements in ordered materials. Neutron scattering experiments at Oak Ridge National Laboratory confirmed this approach can also be used to describe highly disordered materials.
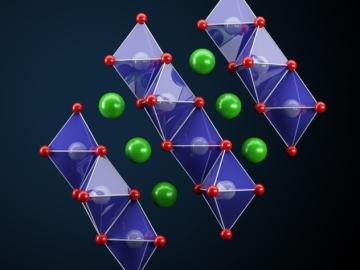
Oak Ridge National Laboratory scientists seeking the source of charge loss in lithium-ion batteries demonstrated that coupling a thin-film cathode with a solid electrolyte is a rapid way to determine the root cause.
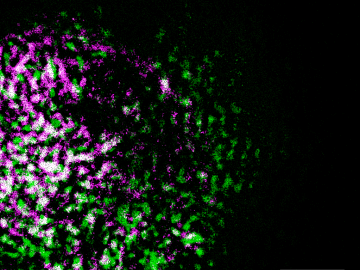
Oak Ridge National Laboratory researchers have built a novel microscope that provides a “chemical lens” for viewing biological systems including cell membranes and biofilms.

Oak Ridge National Laboratory researchers have discovered a better way to separate actinium-227, a rare isotope essential for an FDA-approved cancer treatment.

Oak Ridge National Laboratory researchers have developed a thin film, highly conductive solid-state electrolyte made of a polymer and ceramic-based composite for lithium metal batteries.




