
Filter News
Area of Research
- (-) Materials (17)
- (-) Neutron Science (5)
- Advanced Manufacturing (1)
- Biological Systems (1)
- Biology and Environment (12)
- Clean Energy (34)
- Computer Science (2)
- Electricity and Smart Grid (1)
- Energy Sciences (1)
- Fusion and Fission (1)
- Fusion Energy (1)
- Isotopes (4)
- Materials for Computing (2)
- National Security (3)
- Nuclear Science and Technology (6)
- Quantum information Science (1)
- Sensors and Controls (1)
- Supercomputing (3)
News Topics
- (-) Bioenergy (2)
- (-) Energy Storage (9)
- (-) Isotopes (2)
- (-) Microscopy (6)
- (-) Molten Salt (1)
- (-) Space Exploration (2)
- 3-D Printing/Advanced Manufacturing (6)
- Advanced Reactors (1)
- Artificial Intelligence (1)
- Biomedical (4)
- Buildings (1)
- Chemical Sciences (5)
- Clean Water (1)
- Composites (4)
- Computer Science (1)
- Coronavirus (1)
- Critical Materials (5)
- Decarbonization (1)
- Environment (1)
- Fusion (2)
- Materials (14)
- Materials Science (20)
- Nanotechnology (9)
- Neutron Science (23)
- Nuclear Energy (4)
- Physics (2)
- Polymers (6)
- Quantum Computing (1)
- Quantum Science (2)
- Sustainable Energy (3)
- Transportation (7)
Media Contacts
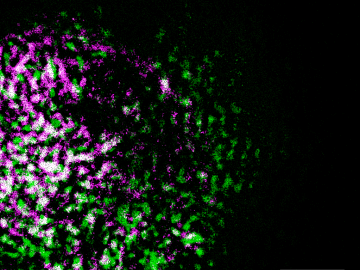
Oak Ridge National Laboratory researchers have built a novel microscope that provides a “chemical lens” for viewing biological systems including cell membranes and biofilms.

Oak Ridge National Laboratory researchers have developed a thin film, highly conductive solid-state electrolyte made of a polymer and ceramic-based composite for lithium metal batteries.
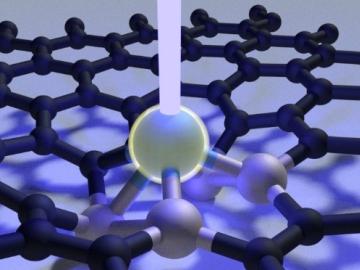
Scientists at Oak Ridge National Laboratory used a focused beam of electrons to stitch platinum-silicon molecules into graphene, marking the first deliberate insertion of artificial molecules into a graphene host matrix.
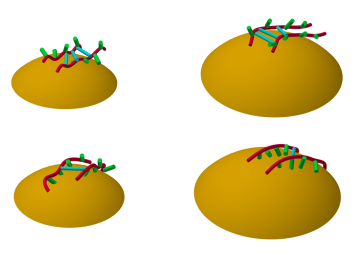
A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.
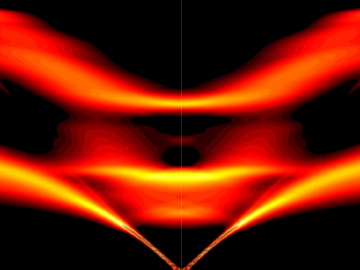
Scientists have discovered a way to alter heat transport in thermoelectric materials, a finding that may ultimately improve energy efficiency as the materials

Oak Ridge National Laboratory scientists analyzed more than 50 years of data showing puzzlingly inconsistent trends about corrosion of structural alloys in molten salts and found one factor mattered most—salt purity.
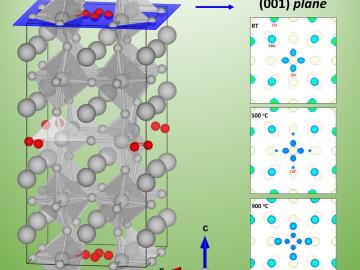
A University of South Carolina research team is investigating the oxygen reduction performance of energy conversion materials called perovskites by using neutron diffraction at Oak Ridge National Laboratory’s Spallation Neutron Source.
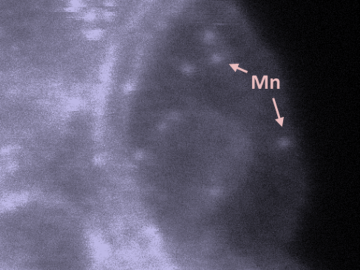
Oak Ridge National Laboratory scientists studying fuel cells as a potential alternative to internal combustion engines used sophisticated electron microscopy to investigate the benefits of replacing high-cost platinum with a lower cost, carbon-nitrogen-manganese-based catalyst.
Physicists turned to the “doubly magic” tin isotope Sn-132, colliding it with a target at Oak Ridge National Laboratory to assess its properties as it lost a neutron to become Sn-131.

An Oak Ridge National Laboratory-led team used a scanning transmission electron microscope to selectively position single atoms below a crystal’s surface for the first time.


