Filter News
Area of Research
- (-) Materials (26)
- (-) Neutron Science (8)
- Advanced Manufacturing (2)
- Biology and Environment (44)
- Clean Energy (36)
- Climate and Environmental Systems (1)
- Computational Engineering (1)
- Computer Science (1)
- Fusion and Fission (2)
- Isotope Development and Production (1)
- Isotopes (14)
- Materials for Computing (7)
- National Security (22)
- Nuclear Science and Technology (3)
- Quantum information Science (1)
- Sensors and Controls (1)
- Supercomputing (30)
News Type
News Topics
- (-) Cybersecurity (4)
- (-) Environment (11)
- (-) Isotopes (5)
- (-) Machine Learning (2)
- (-) Polymers (8)
- (-) Security (2)
- (-) Space Exploration (1)
- (-) Summit (4)
- 3-D Printing/Advanced Manufacturing (15)
- Advanced Reactors (1)
- Artificial Intelligence (4)
- Big Data (1)
- Bioenergy (10)
- Biology (8)
- Biomedical (7)
- Biotechnology (1)
- Buildings (2)
- Chemical Sciences (22)
- Climate Change (5)
- Composites (3)
- Computer Science (11)
- Coronavirus (7)
- Critical Materials (8)
- Decarbonization (5)
- Energy Storage (21)
- Exascale Computing (1)
- Frontier (3)
- Fusion (4)
- Grid (2)
- High-Performance Computing (3)
- ITER (1)
- Materials (43)
- Materials Science (45)
- Microscopy (15)
- Molten Salt (2)
- Nanotechnology (26)
- National Security (3)
- Net Zero (1)
- Neutron Science (49)
- Nuclear Energy (5)
- Partnerships (8)
- Physics (22)
- Quantum Computing (2)
- Quantum Science (11)
- Renewable Energy (1)
- Sustainable Energy (8)
- Transformational Challenge Reactor (1)
- Transportation (7)
Media Contacts

Real-time measurements captured by researchers at ORNL provide missing insight into chemical separations to recover cobalt, a critical raw material used to make batteries and magnets for modern technologies.

Scientists at ORNL used neutron scattering and supercomputing to better understand how an organic solvent and water work together to break down plant biomass, creating a pathway to significantly improve the production of renewable
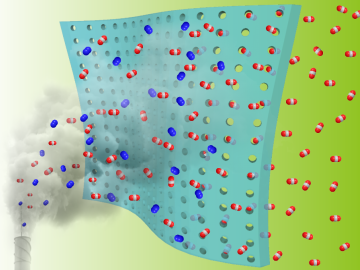
Researchers at the Department of Energy’s Oak Ridge National Laboratory and the University of Tennessee, Knoxville, are advancing gas membrane materials to expand practical technology options for reducing industrial carbon emissions.
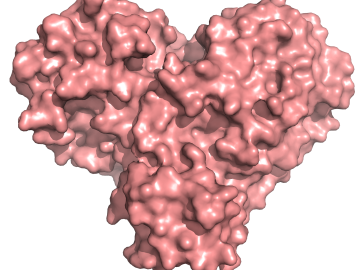
A team of researchers has performed the first room-temperature X-ray measurements on the SARS-CoV-2 main protease — the enzyme that enables the virus to reproduce.

OAK RIDGE, Tenn., Feb. 27, 2020 — Researchers at Oak Ridge National Laboratory and the University of Tennessee achieved a rare look at the inner workings of polymer self-assembly at an oil-water interface to advance materials for neuromorphic computing and bio-inspired technologies.
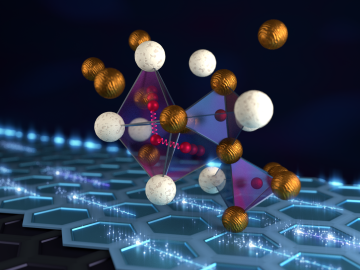
An international team of researchers has discovered the hydrogen atoms in a metal hydride material are much more tightly spaced than had been predicted for decades — a feature that could possibly facilitate superconductivity at or near room temperature and pressure.
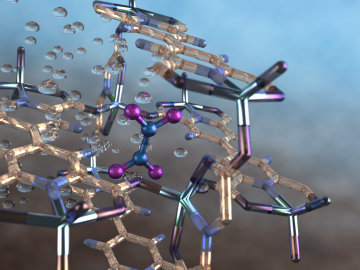
An international team of scientists, led by the University of Manchester, has developed a metal-organic framework, or MOF, material

Researchers at the Department of Energy’s Oak Ridge National Laboratory have received five 2019 R&D 100 Awards, increasing the lab’s total to 221 since the award’s inception in 1963.
OAK RIDGE, Tenn., March 1, 2019—ReactWell, LLC, has licensed a novel waste-to-fuel technology from the Department of Energy’s Oak Ridge National Laboratory to improve energy conversion methods for cleaner, more efficient oil and gas, chemical and

Vera Bocharova at the Department of Energy’s Oak Ridge National Laboratory investigates the structure and dynamics of soft materials—polymer nanocomposites, polymer electrolytes and biological macromolecules—to advance materials and technologies for energy, medicine and other applications.




