Filter News
Area of Research
- (-) Materials (43)
- Advanced Manufacturing (4)
- Biology and Environment (9)
- Clean Energy (43)
- Computational Engineering (2)
- Computer Science (4)
- Electricity and Smart Grid (1)
- Energy Sciences (1)
- Functional Materials for Energy (2)
- Fusion and Fission (10)
- Fusion Energy (6)
- Isotopes (2)
- Materials Characterization (1)
- Materials for Computing (10)
- Materials Under Extremes (1)
- Mathematics (1)
- National Security (3)
- Neutron Science (10)
- Nuclear Science and Technology (5)
- Supercomputing (9)
- Transportation Systems (1)
News Type
News Topics
- (-) Clean Water (1)
- (-) Energy Storage (16)
- (-) Fusion (4)
- (-) Machine Learning (2)
- (-) Materials Science (31)
- (-) Molten Salt (2)
- 3-D Printing/Advanced Manufacturing (12)
- Advanced Reactors (3)
- Artificial Intelligence (3)
- Bioenergy (5)
- Biology (2)
- Biomedical (2)
- Buildings (2)
- Chemical Sciences (11)
- Climate Change (3)
- Composites (5)
- Computer Science (9)
- Coronavirus (1)
- Critical Materials (6)
- Cybersecurity (3)
- Decarbonization (2)
- Environment (5)
- Frontier (2)
- Grid (2)
- High-Performance Computing (2)
- Irradiation (1)
- Isotopes (4)
- ITER (1)
- Materials (35)
- Microscopy (14)
- Nanotechnology (17)
- National Security (1)
- Neutron Science (8)
- Nuclear Energy (5)
- Partnerships (2)
- Physics (8)
- Polymers (8)
- Quantum Computing (2)
- Quantum Science (6)
- Simulation (1)
- Space Exploration (1)
- Sustainable Energy (6)
- Transformational Challenge Reactor (1)
- Transportation (9)
Media Contacts

Electric vehicles can drive longer distances if their lithium-ion batteries deliver more energy in a lighter package. A prime weight-loss candidate is the current collector, a component that often adds 10% to the weight of a battery cell without contributing energy.

ORNL scientists found that a small tweak created big performance improvements in a type of solid-state battery, a technology considered vital to broader electric vehicle adoption.

Warming a crystal of the mineral fresnoite, ORNL scientists discovered that excitations called phasons carried heat three times farther and faster than phonons, the excitations that usually carry heat through a material.

Zheng Gai, a senior staff scientist at ORNL’s Center for Nanophase Materials Sciences, has been selected as editor-in-chief of the Spin Crossover and Spintronics section of Magnetochemistry.

Anne Campbell, an R&D associate in ORNL’s Materials Science and Technology Division since 2016, has been selected as an associate editor of the Journal of Nuclear Materials.

Seven scientists at the Department of Energy’s Oak Ridge National Laboratory have been named Battelle Distinguished Inventors, in recognition of their obtaining 14 or more patents during their careers at the lab.

Three researchers at ORNL have been named ORNL Corporate Fellows in recognition of significant career accomplishments and continued leadership in their scientific fields.

Oak Ridge National Laboratory researchers serendipitously discovered when they automated the beam of an electron microscope to precisely drill holes in the atomically thin lattice of graphene, the drilled holes closed up.

Sheng Dai, a Corporate Fellow and section head at the Department of Energy’s Oak Ridge National Laboratory, has been selected by the DOE Office of Science as a 2023 Distinguished Scientist Fellow.
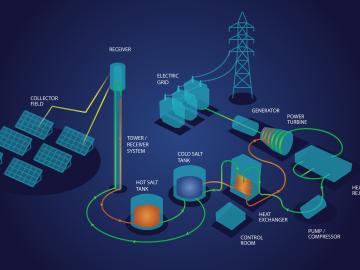
Oak Ridge National Laboratory scientists recently demonstrated a low-temperature, safe route to purifying molten chloride salts that minimizes their ability to corrode metals. This method could make the salts useful for storing energy generated from the sun’s heat.




