Filter News
Area of Research
- (-) Materials (21)
- Advanced Manufacturing (2)
- Biology and Environment (13)
- Clean Energy (16)
- Computational Engineering (1)
- Computer Science (3)
- Electricity and Smart Grid (1)
- Functional Materials for Energy (1)
- Fusion and Fission (1)
- Isotopes (14)
- Materials for Computing (3)
- National Security (19)
- Neutron Science (10)
- Nuclear Science and Technology (3)
- Quantum information Science (2)
- Supercomputing (36)
News Topics
- (-) Cybersecurity (3)
- (-) Isotopes (5)
- (-) Machine Learning (3)
- (-) Quantum Science (10)
- (-) Summit (1)
- 3-D Printing/Advanced Manufacturing (13)
- Advanced Reactors (2)
- Artificial Intelligence (5)
- Bioenergy (9)
- Biology (4)
- Biomedical (3)
- Buildings (3)
- Chemical Sciences (22)
- Climate Change (5)
- Composites (3)
- Computer Science (8)
- Coronavirus (2)
- Critical Materials (8)
- Decarbonization (5)
- Energy Storage (21)
- Environment (9)
- Exascale Computing (1)
- Frontier (3)
- Fusion (3)
- Grid (3)
- High-Performance Computing (3)
- Irradiation (1)
- ITER (1)
- Materials (46)
- Materials Science (43)
- Microscopy (17)
- Molten Salt (2)
- Nanotechnology (25)
- National Security (3)
- Net Zero (1)
- Neutron Science (21)
- Nuclear Energy (5)
- Partnerships (9)
- Physics (20)
- Polymers (8)
- Quantum Computing (2)
- Renewable Energy (1)
- Security (1)
- Simulation (1)
- Sustainable Energy (8)
- Transformational Challenge Reactor (1)
- Transportation (5)
Media Contacts

An international multi-institution team of scientists has synthesized graphene nanoribbons – ultrathin strips of carbon atoms – on a titanium dioxide surface using an atomically precise method that removes a barrier for custom-designed carbon

Researchers at ORNL used quantum optics to advance state-of-the-art microscopy and illuminate a path to detecting material properties with greater sensitivity than is possible with traditional tools.

Five researchers at the Department of Energy’s Oak Ridge National Laboratory have been named ORNL Corporate Fellows in recognition of significant career accomplishments and continued leadership in their scientific fields.
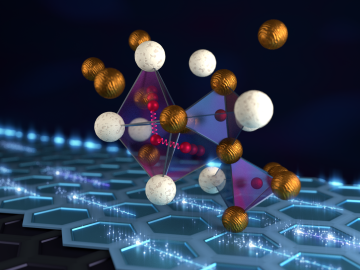
A team led by the Department of Energy’s Oak Ridge National Laboratory synthesized a tiny structure with high surface area and discovered how its unique architecture drives ions across interfaces to transport energy or information.
An international team of researchers has discovered the hydrogen atoms in a metal hydride material are much more tightly spaced than had been predicted for decades — a feature that could possibly facilitate superconductivity at or near room temperature and pressure.

Researchers at the Department of Energy’s Oak Ridge National Laboratory have received five 2019 R&D 100 Awards, increasing the lab’s total to 221 since the award’s inception in 1963.

A team led by scientists at the Department of Energy’s Oak Ridge National Laboratory explored how atomically thin two-dimensional (2D) crystals can grow over 3D objects and how the curvature of those objects can stretch and strain the

OAK RIDGE, Tenn., May 7, 2019—Energy Secretary Rick Perry, Congressman Chuck Fleischmann and lab officials today broke ground on a multipurpose research facility that will provide state-of-the-art laboratory space

OAK RIDGE, Tenn., Jan. 31, 2019—A new electron microscopy technique that detects the subtle changes in the weight of proteins at the nanoscale—while keeping the sample intact—could open a new pathway for deeper, more comprehensive studies of the basic building blocks of life.
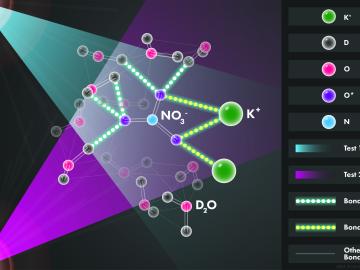
Scientists at the Department of Energy’s Oak Ridge National Laboratory used neutrons, isotopes and simulations to “see” the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come

