Filter News
Area of Research
- (-) Neutron Science (20)
- Biological Systems (1)
- Biology and Environment (62)
- Biology and Soft Matter (1)
- Clean Energy (77)
- Climate and Environmental Systems (1)
- Computational Biology (1)
- Computational Engineering (1)
- Computer Science (1)
- Fuel Cycle Science and Technology (1)
- Fusion and Fission (9)
- Fusion Energy (1)
- Isotopes (6)
- Materials (44)
- Materials for Computing (10)
- National Security (40)
- Nuclear Science and Technology (8)
- Supercomputing (70)
News Topics
- (-) Big Data (2)
- (-) Biomedical (9)
- (-) Climate Change (1)
- (-) Coronavirus (8)
- (-) Energy Storage (4)
- (-) Frontier (1)
- (-) National Security (2)
- (-) Polymers (1)
- 3-D Printing/Advanced Manufacturing (6)
- Artificial Intelligence (5)
- Bioenergy (5)
- Biology (5)
- Biotechnology (1)
- Chemical Sciences (1)
- Clean Water (2)
- Composites (1)
- Computer Science (13)
- Cybersecurity (1)
- Decarbonization (2)
- Environment (6)
- Fossil Energy (1)
- Fusion (1)
- High-Performance Computing (2)
- Machine Learning (3)
- Materials (11)
- Materials Science (20)
- Mathematics (1)
- Microscopy (2)
- Nanotechnology (8)
- Neutron Science (73)
- Nuclear Energy (2)
- Physics (8)
- Quantum Computing (1)
- Quantum Science (5)
- Security (2)
- Space Exploration (2)
- Summit (6)
- Sustainable Energy (2)
- Transportation (3)
Media Contacts
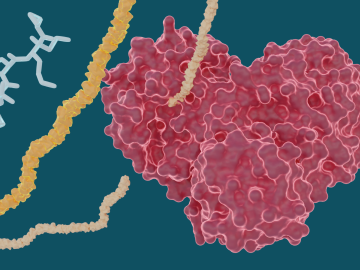
Experiments led by researchers at ORNL have determined that several hepatitis C drugs can inhibit the SARS-CoV-2 main protease, a crucial protein enzyme that enables the novel coronavirus to reproduce.
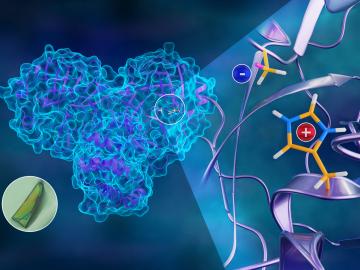
To better understand how the novel coronavirus behaves and how it can be stopped, scientists have completed a three-dimensional map that reveals the location of every atom in an enzyme molecule critical to SARS-CoV-2 reproduction.
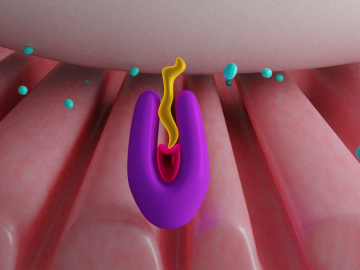
Pick your poison. It can be deadly for good reasons such as protecting crops from harmful insects or fighting parasite infection as medicine — or for evil as a weapon for bioterrorism. Or, in extremely diluted amounts, it can be used to enhance beauty.
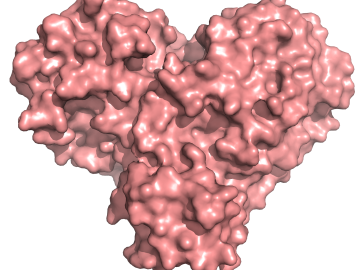
A team of researchers has performed the first room-temperature X-ray measurements on the SARS-CoV-2 main protease — the enzyme that enables the virus to reproduce.

COVID-19 has upended nearly every aspect of our daily lives and forced us all to rethink how we can continue our work in a more physically isolated world.
In the race to identify solutions to the COVID-19 pandemic, researchers at the Department of Energy’s Oak Ridge National Laboratory are joining the fight by applying expertise in computational science, advanced manufacturing, data science and neutron science.

Biological membranes, such as the “walls” of most types of living cells, primarily consist of a double layer of lipids, or “lipid bilayer,” that forms the structure, and a variety of embedded and attached proteins with highly specialized functions, including proteins that rapidly and selectively transport ions and molecules in and out of the cell.

Two of the researchers who share the Nobel Prize in Chemistry announced Wednesday—John B. Goodenough of the University of Texas at Austin and M. Stanley Whittingham of Binghamton University in New York—have research ties to ORNL.
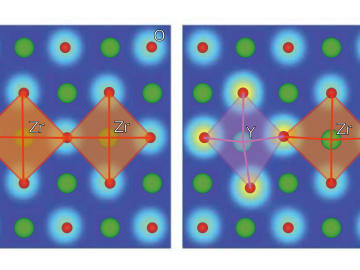
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.
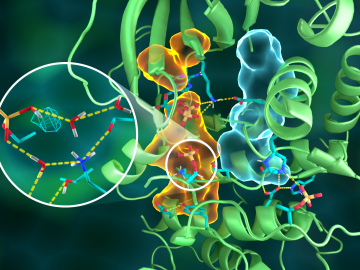
OAK RIDGE, Tenn., March 20, 2019—Direct observations of the structure and catalytic mechanism of a prototypical kinase enzyme—protein kinase A or PKA—will provide researchers and drug developers with significantly enhanced abilities to understand and treat fatal diseases and neurological disorders such as cancer, diabetes, and cystic fibrosis.




