Filter News
Area of Research
News Topics
- (-) 3-D Printing/Advanced Manufacturing (1)
- (-) Advanced Reactors (1)
- (-) Bioenergy (1)
- (-) Biomedical (2)
- (-) Space Exploration (1)
- Artificial Intelligence (1)
- Big Data (1)
- Biology (1)
- Biotechnology (1)
- Clean Water (2)
- Computer Science (2)
- Coronavirus (1)
- Decarbonization (1)
- Energy Storage (2)
- Environment (4)
- Fossil Energy (1)
- High-Performance Computing (1)
- Machine Learning (2)
- Materials (5)
- Materials Science (4)
- Nanotechnology (2)
- Neutron Science (30)
- Nuclear Energy (5)
- Physics (2)
- Sustainable Energy (1)
Media Contacts

How do you get water to float in midair? With a WAND2, of course. But it’s hardly magic. In fact, it’s a scientific device used by scientists to study matter.

The Department of Energy’s Office of Science has selected three ORNL research teams to receive funding through DOE’s new Biopreparedness Research Virtual Environment initiative.

How did we get from stardust to where we are today? That’s the question NASA scientist Andrew Needham has pondered his entire career.
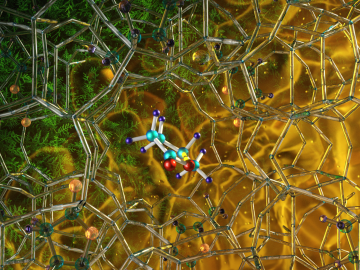
Illustration of the optimized zeolite catalyst, or NbAlS-1, which enables a highly efficient chemical reaction to create butene, a renewable source of energy, without expending high amounts of energy for the conversion. Credit: Jill Hemman, Oak Ridge National Laboratory/U.S. Dept. of Energy

Ask Tyler Gerczak to find a negative in working at the Department of Energy’s Oak Ridge National Laboratory, and his only complaint is the summer weather. It is not as forgiving as the summers in Pulaski, Wisconsin, his hometown.
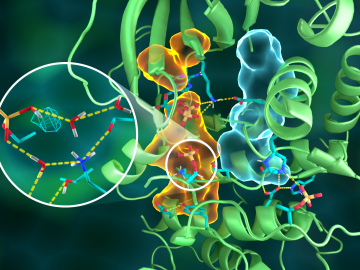
OAK RIDGE, Tenn., March 20, 2019—Direct observations of the structure and catalytic mechanism of a prototypical kinase enzyme—protein kinase A or PKA—will provide researchers and drug developers with significantly enhanced abilities to understand and treat fatal diseases and neurological disorders such as cancer, diabetes, and cystic fibrosis.




