Filter News
Area of Research
- (-) Biology and Environment (7)
- (-) Materials for Computing (2)
- (-) Neutron Science (13)
- Advanced Manufacturing (4)
- Clean Energy (22)
- Computer Science (1)
- Fusion Energy (2)
- Isotopes (1)
- Materials (43)
- National Security (4)
- Nuclear Science and Technology (2)
- Supercomputing (16)
- Transportation Systems (1)
News Type
News Topics
- (-) Bioenergy (3)
- (-) Biomedical (11)
- (-) Materials Science (11)
- 3-D Printing/Advanced Manufacturing (4)
- Artificial Intelligence (1)
- Big Data (1)
- Biology (3)
- Chemical Sciences (2)
- Climate Change (4)
- Computer Science (10)
- Coronavirus (7)
- Energy Storage (4)
- Environment (9)
- High-Performance Computing (1)
- Isotopes (1)
- Machine Learning (1)
- Materials (2)
- Microscopy (2)
- Molten Salt (1)
- Nanotechnology (8)
- National Security (1)
- Neutron Science (32)
- Nuclear Energy (1)
- Physics (5)
- Polymers (1)
- Quantum Science (3)
- Summit (7)
- Sustainable Energy (5)
- Transportation (3)
Media Contacts
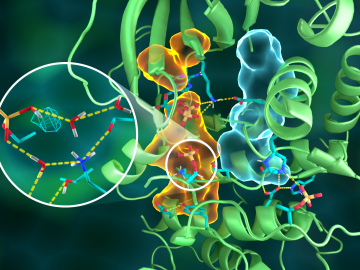
OAK RIDGE, Tenn., March 20, 2019—Direct observations of the structure and catalytic mechanism of a prototypical kinase enzyme—protein kinase A or PKA—will provide researchers and drug developers with significantly enhanced abilities to understand and treat fatal diseases and neurological disorders such as cancer, diabetes, and cystic fibrosis.
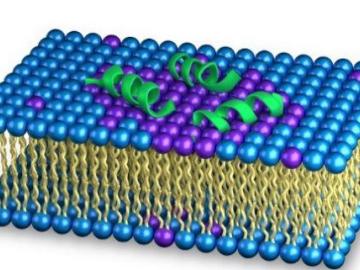
As the rise of antibiotic-resistant bacteria known as superbugs threatens public health, Oak Ridge National Laboratory’s Shuo Qian and Veerendra Sharma from the Bhaba Atomic Research Centre in India are using neutron scattering to study how an antibacterial peptide interacts with and fights harmful bacteria.

