Filter News
Area of Research
News Topics
- (-) Clean Water (5)
- (-) Cybersecurity (5)
- (-) Energy Storage (8)
- (-) Nanotechnology (6)
- (-) Physics (6)
- (-) Polymers (2)
- 3-D Printing/Advanced Manufacturing (15)
- Advanced Reactors (7)
- Artificial Intelligence (12)
- Big Data (7)
- Bioenergy (9)
- Biomedical (5)
- Biotechnology (1)
- Composites (2)
- Computer Science (35)
- Environment (19)
- Exascale Computing (2)
- Frontier (2)
- Fusion (5)
- Grid (5)
- Isotopes (1)
- Machine Learning (5)
- Materials Science (20)
- Mercury (1)
- Microscopy (5)
- Molten Salt (1)
- Neutron Science (18)
- Nuclear Energy (17)
- Quantum Science (10)
- Security (2)
- Space Exploration (4)
- Summit (9)
- Sustainable Energy (8)
- Transportation (12)
Media Contacts
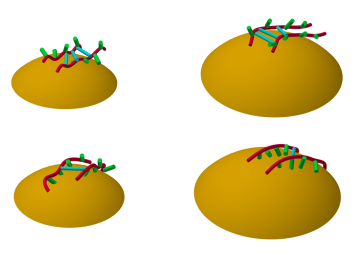
A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.

Scientists at Oak Ridge National Laboratory studying quantum communications have discovered a more practical way to share secret messages among three parties, which could ultimately lead to better cybersecurity for the electric grid
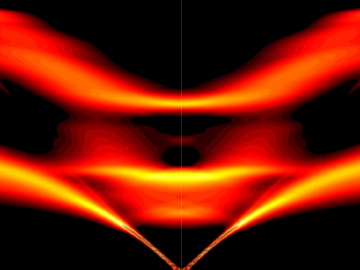
Scientists have discovered a way to alter heat transport in thermoelectric materials, a finding that may ultimately improve energy efficiency as the materials

An ORNL-led team's observation of certain crystalline ice phases challenges accepted theories about super-cooled water and non-crystalline ice. Their findings, reported in the journal Nature, will also lead to better understanding of ice and its various phases found on other planets, moons and elsewhere in space.

Researchers at the Department of Energy’s Oak Ridge National Laboratory, Pacific Northwest National Laboratory and Washington State University teamed up to investigate the complex dynamics of low-water liquids that challenge nuclear waste processing at federal cleanup sites.
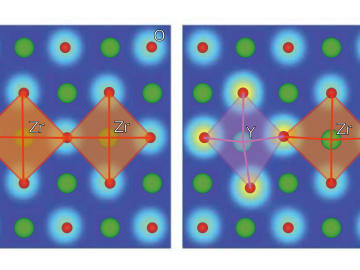
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.
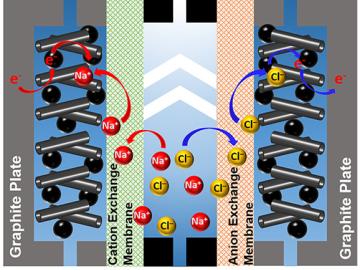
A team of scientists led by Oak Ridge National Laboratory used carbon nanotubes to improve a desalination process that attracts and removes ionic compounds such as salt from water using charged electrodes.

OAK RIDGE, Tenn., March 11, 2019—An international collaboration including scientists at the Department of Energy’s Oak Ridge National Laboratory solved a 50-year-old puzzle that explains why beta decays of atomic nuclei

Vera Bocharova at the Department of Energy’s Oak Ridge National Laboratory investigates the structure and dynamics of soft materials—polymer nanocomposites, polymer electrolytes and biological macromolecules—to advance materials and technologies for energy, medicine and other applications.




