Filter News
Area of Research
- (-) Neutron Science (13)
- Advanced Manufacturing (1)
- Biological Systems (1)
- Biology and Environment (26)
- Building Technologies (1)
- Clean Energy (49)
- Climate and Environmental Systems (1)
- Computational Engineering (1)
- Computer Science (10)
- Energy Sciences (1)
- Fusion and Fission (3)
- Fusion Energy (1)
- Isotopes (1)
- Materials (33)
- National Security (19)
- Nuclear Science and Technology (2)
- Quantum information Science (3)
- Sensors and Controls (1)
- Supercomputing (53)
News Type
News Topics
- (-) Artificial Intelligence (1)
- (-) Biomedical (4)
- (-) Clean Water (2)
- (-) Computer Science (2)
- (-) Energy Storage (4)
- (-) Machine Learning (2)
- (-) Microscopy (1)
- (-) Sustainable Energy (1)
- 3-D Printing/Advanced Manufacturing (2)
- Big Data (1)
- Bioenergy (3)
- Biology (1)
- Biotechnology (1)
- Chemical Sciences (1)
- Composites (1)
- Coronavirus (1)
- Decarbonization (1)
- Environment (5)
- Fossil Energy (1)
- High-Performance Computing (1)
- Materials (7)
- Materials Science (6)
- Nanotechnology (3)
- Neutron Science (46)
- Nuclear Energy (2)
- Physics (4)
- Quantum Science (1)
- Space Exploration (1)
- Transportation (2)
Media Contacts

How do you get water to float in midair? With a WAND2, of course. But it’s hardly magic. In fact, it’s a scientific device used by scientists to study matter.
Currently, the biggest hurdle for electric vehicles, or EVs, is the development of advanced battery technology to extend driving range, safety and reliability.

Neutron experiments can take days to complete, requiring researchers to work long shifts to monitor progress and make necessary adjustments. But thanks to advances in artificial intelligence and machine learning, experiments can now be done remotely and in half the time.

Like most scientists, Chengping Chai is not content with the surface of things: He wants to probe beyond to learn what’s really going on. But in his case, he is literally building a map of the world beneath, using seismic and acoustic data that reveal when and where the earth moves.

ORNL computer scientist Catherine Schuman returned to her alma mater, Harriman High School, to lead Hour of Code activities and talk to students about her job as a researcher.

Two of the researchers who share the Nobel Prize in Chemistry announced Wednesday—John B. Goodenough of the University of Texas at Austin and M. Stanley Whittingham of Binghamton University in New York—have research ties to ORNL.
Scientists have discovered a way to alter heat transport in thermoelectric materials, a finding that may ultimately improve energy efficiency as the materials

Researchers at the Department of Energy’s Oak Ridge National Laboratory, Pacific Northwest National Laboratory and Washington State University teamed up to investigate the complex dynamics of low-water liquids that challenge nuclear waste processing at federal cleanup sites.
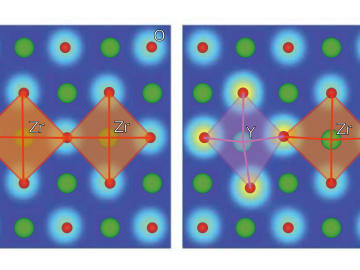
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.
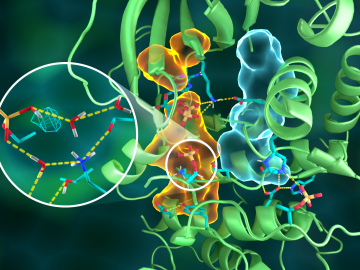
OAK RIDGE, Tenn., March 20, 2019—Direct observations of the structure and catalytic mechanism of a prototypical kinase enzyme—protein kinase A or PKA—will provide researchers and drug developers with significantly enhanced abilities to understand and treat fatal diseases and neurological disorders such as cancer, diabetes, and cystic fibrosis.