Filter News
Area of Research
- (-) Neutron Science (20)
- Advanced Manufacturing (4)
- Biological Systems (1)
- Biology and Environment (8)
- Clean Energy (55)
- Climate and Environmental Systems (1)
- Computer Science (2)
- Energy Sciences (1)
- Fusion and Fission (2)
- Fusion Energy (8)
- Materials (61)
- Materials for Computing (4)
- National Security (5)
- Nuclear Science and Technology (12)
- Nuclear Systems Modeling, Simulation and Validation (1)
- Quantum information Science (3)
- Supercomputing (21)
- Transportation Systems (1)
News Topics
- (-) Bioenergy (4)
- (-) Climate Change (1)
- (-) Energy Storage (3)
- (-) Materials Science (10)
- (-) Nanotechnology (4)
- (-) Physics (5)
- 3-D Printing/Advanced Manufacturing (2)
- Artificial Intelligence (1)
- Big Data (1)
- Biomedical (7)
- Clean Water (1)
- Computer Science (8)
- Coronavirus (5)
- Environment (5)
- Machine Learning (2)
- Mathematics (1)
- Microscopy (2)
- National Security (1)
- Neutron Science (43)
- Nuclear Energy (2)
- Polymers (1)
- Quantum Science (3)
- Security (1)
- Summit (5)
- Sustainable Energy (1)
- Transportation (2)
Media Contacts

Biological membranes, such as the “walls” of most types of living cells, primarily consist of a double layer of lipids, or “lipid bilayer,” that forms the structure, and a variety of embedded and attached proteins with highly specialized functions, including proteins that rapidly and selectively transport ions and molecules in and out of the cell.
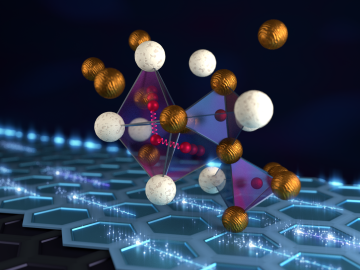
An international team of researchers has discovered the hydrogen atoms in a metal hydride material are much more tightly spaced than had been predicted for decades — a feature that could possibly facilitate superconductivity at or near room temperature and pressure.
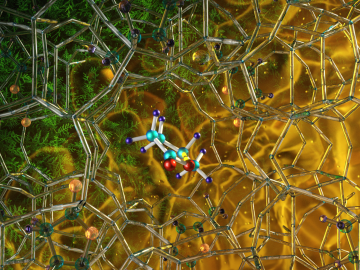
Illustration of the optimized zeolite catalyst, or NbAlS-1, which enables a highly efficient chemical reaction to create butene, a renewable source of energy, without expending high amounts of energy for the conversion. Credit: Jill Hemman, Oak Ridge National Laboratory/U.S. Dept. of Energy

Scientists at the U.S. Department of Energy’s Brookhaven National Laboratory have new experimental evidence and a predictive theory that solves a long-standing materials science mystery: why certain crystalline materials shrink when heated.

Two of the researchers who share the Nobel Prize in Chemistry announced Wednesday—John B. Goodenough of the University of Texas at Austin and M. Stanley Whittingham of Binghamton University in New York—have research ties to ORNL.
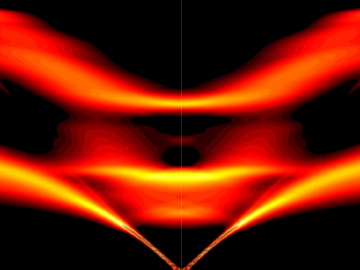
Scientists have discovered a way to alter heat transport in thermoelectric materials, a finding that may ultimately improve energy efficiency as the materials

An ORNL-led team's observation of certain crystalline ice phases challenges accepted theories about super-cooled water and non-crystalline ice. Their findings, reported in the journal Nature, will also lead to better understanding of ice and its various phases found on other planets, moons and elsewhere in space.
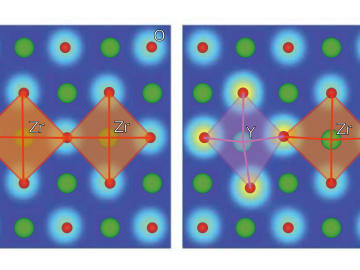
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.
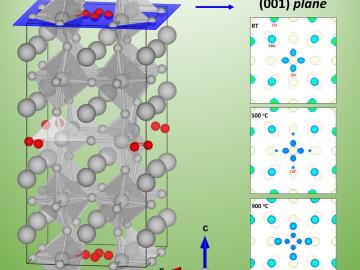
A University of South Carolina research team is investigating the oxygen reduction performance of energy conversion materials called perovskites by using neutron diffraction at Oak Ridge National Laboratory’s Spallation Neutron Source.

After more than a year of operation at the Department of Energy’s (DOE’s) Oak Ridge National Laboratory (ORNL), the COHERENT experiment, using the world’s smallest neutrino detector, has found a big fingerprint of the elusive, electrically neutral particles that interact only weakly with matter.

