Scientists at the Department of Energy’s Oak Ridge National Laboratory and the University of Tennessee, Knoxville, have found a way to simultaneously increase the strength and ductility of an alloy by introducing tiny precipitates into its matrix and tuning their size and spacing. The precipitates are solids that separate from the metal mixture as the alloy cools. The results, published in the journal Nature, will open new avenues for advancing structural materials.
Ductility is a measure of a material’s ability to undergo permanent deformation without breaking. It determines, among other things, how much a material can elongate before fracturing and whether that fracturing will be graceful or catastrophic. The higher the strength and ductility, the tougher the material.
“A holy grail of structural materials has long been, how do you simultaneously enhance strength and ductility?” said Easo George, principal investigator of the study and Governor’s Chair for Advanced Alloy Theory and Development at ORNL and UT. “Defeating the strength–ductility trade-off will enable a new generation of lightweight, strong, damage-tolerant materials.”
If structural materials could become stronger and more ductile, components of cars, planes, power plants, buildings and bridges could be built using less material. Lighter-weight vehicles would be more energy-efficient to make and operate, and tougher infrastructure would be more resilient.
Co-principal investigator Ying Yang of ORNL conceived and led the Nature study. Guided by computational thermodynamics simulations, she designed and custom-made model alloys with the special ability to undergo a phase transformation from a face-centered cubic, or FCC, to a body-centered cubic, or BCC, crystal structure, driven by changes in either temperature or stress.
“We put nanoprecipitates into a transformable matrix and carefully controlled their attributes, which in turn controlled when and how the matrix transformed,” Yang said. “In this material, we intentionally induced the matrix to have the capability to undergo a phase transformation.”
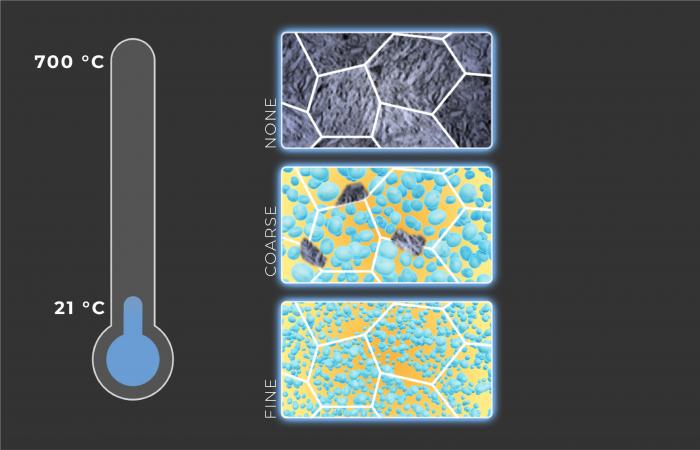
The alloy contains four major elements — iron, nickel, aluminum and titanium — that form the matrix and precipitates, and three minor elements — carbon, zirconium and boron — that limit the size of grains, individual metallic crystals.
The researchers carefully kept the composition of the matrix and the total amount of nanoprecipitates the same in different samples. However, they varied precipitate sizes and spacings by adjusting the processing temperature and time. For comparison, a reference alloy without precipitates but having the same composition as the matrix of the precipitate-containing alloy was also prepared and tested.
“The strength of a material usually depends on how close the precipitates are to each other,” George said. “When you make them a few nanometers [billionths of a meter] in size, they can be very closely spaced. The more closely spaced they are, the stronger the material gets.”
While nanoprecipitates in conventional alloys can make them super strong, they also make the alloys very brittle. The team’s alloy avoids this brittleness because the precipitates perform a second useful function: by spatially constraining the matrix, they prevent it from transforming during a thermal quench, a quick immersion in water that cools the alloy to room temperature. Consequently, the matrix remains in a metastable FCC state. When the alloy is then stretched (“strained”), it progressively transforms from metastable FCC to stable BCC. This phase transformation during straining increases strength while maintaining adequate ductility. In contrast, the alloy without precipitates transforms fully to stable FCC during the thermal quench, which precludes further transformation during straining. As a result, it is both weaker and more brittle than the alloy with precipitates. Together, the complementary mechanisms of conventional precipitation strengthening and deformation-induced transformation increased strength by 20%-90% and elongation by 300%.
“Adding precipitates to block dislocations and make materials ultra-strong is well known,” George said. “What is new here is that adjusting the spacing of these precipitates also affects phase transformation propensity, which allows multiple deformation mechanisms to be activated as needed to enhance ductility.”
The study also revealed a surprising reversal of the normal strengthening effect of nanoprecipitates: an alloy with coarse, widely spaced precipitates is stronger than the same alloy with fine, closely spaced precipitates. This reversal happens when the nanoprecipitates become so tiny and tightly packed that the phase transformation is essentially shut down during straining of the material, not unlike the transformation suppressed during the thermal quench.
This study relied on complementary techniques performed at DOE Office of Science user facilities at ORNL to characterize the nanoprecipitates and deformation mechanisms. At the Center for Nanophase Materials Sciences, atom probe tomography showed the size, distribution and chemical composition of precipitates, whereas transmission electron microscopy exposed atomistic details of local regions. At the High Flux Isotope Reactor, small-angle neutron scattering quantified the distribution of fine precipitates. And at the Spallation Neutron Source, neutron diffraction probed the phase transformation after different levels of strain.
“This research introduces a new family of structural alloys,” Yang said. “Precipitate characteristics and alloy chemistry can be precisely tailored to activate deformation mechanisms exactly when needed to thwart the strength-ductility trade-off.”
Next the team will investigate additional factors and deformation mechanisms to identify combinations that could further improve mechanical properties.
It turns out, there is a lot of room for improvement. “Today’s structural materials realize but a small fraction — perhaps only 10% — of their theoretically capable strengths,” George said. “Imagine the weight savings that would be possible in a car or an airplane — and the consequent energy savings — if this strength could be doubled or tripled while maintaining adequate ductility.”
The title of the Nature paper is “Bifunctional nanoprecipitates strengthen and ductilize a medium-entropy alloy.”
The DOE Office of Science and ORNL’s Laboratory Directed Research and Development Program supported the research.
UT-Battelle manages ORNL for the Department of Energy’s Office of Science, the single largest supporter of basic research in the physical sciences in the United States. The Office of Science is