Filter News
Area of Research
News Topics
- (-) Isotopes (52)
- (-) ITER (7)
- (-) Mercury (12)
- 3-D Printing/Advanced Manufacturing (119)
- Advanced Reactors (34)
- Artificial Intelligence (91)
- Big Data (53)
- Bioenergy (91)
- Biology (98)
- Biomedical (58)
- Biotechnology (22)
- Buildings (57)
- Chemical Sciences (63)
- Clean Water (29)
- Climate Change (99)
- Composites (26)
- Computer Science (186)
- Coronavirus (46)
- Critical Materials (25)
- Cybersecurity (35)
- Decarbonization (78)
- Education (4)
- Element Discovery (1)
- Emergency (2)
- Energy Storage (108)
- Environment (194)
- Exascale Computing (37)
- Fossil Energy (5)
- Frontier (42)
- Fusion (53)
- Grid (62)
- High-Performance Computing (84)
- Hydropower (11)
- Irradiation (3)
- Machine Learning (47)
- Materials (143)
- Materials Science (138)
- Mathematics (7)
- Microelectronics (2)
- Microscopy (51)
- Molten Salt (8)
- Nanotechnology (60)
- National Security (60)
- Net Zero (13)
- Neutron Science (130)
- Nuclear Energy (106)
- Partnerships (42)
- Physics (59)
- Polymers (33)
- Quantum Computing (32)
- Quantum Science (67)
- Renewable Energy (2)
- Security (24)
- Simulation (46)
- Software (1)
- Space Exploration (25)
- Statistics (3)
- Summit (57)
- Sustainable Energy (125)
- Transformational Challenge Reactor (7)
- Transportation (95)
Media Contacts

OAK RIDGE, Tenn., Jan. 31, 2019—A new electron microscopy technique that detects the subtle changes in the weight of proteins at the nanoscale—while keeping the sample intact—could open a new pathway for deeper, more comprehensive studies of the basic building blocks of life.
Physicists turned to the “doubly magic” tin isotope Sn-132, colliding it with a target at Oak Ridge National Laboratory to assess its properties as it lost a neutron to become Sn-131.
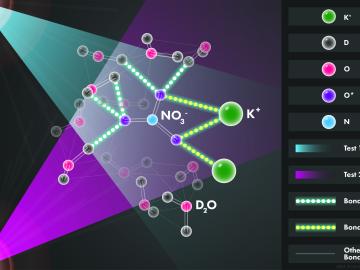
Scientists at the Department of Energy’s Oak Ridge National Laboratory used neutrons, isotopes and simulations to “see” the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come

Biologists from Oak Ridge National Laboratory and the Smithsonian Environmental Research Center have confirmed that microorganisms called methanogens can transform mercury into the neurotoxin methylmercury with varying efficiency across species.

A tiny vial of gray powder produced at the Department of Energy’s Oak Ridge National Laboratory is the backbone of a new experiment to study the intense magnetic fields created in nuclear collisions.

The Department of Energy’s Oak Ridge National Laboratory is now producing actinium-227 (Ac-227) to meet projected demand for a highly effective cancer drug through a 10-year contract between the U.S. DOE Isotope Program and Bayer.

“Made in the USA.” That can now be said of the radioactive isotope molybdenum-99 (Mo-99), last made in the United States in the late 1980s. Its short-lived decay product, technetium-99m (Tc-99m), is the most widely used radioisotope in medical diagnostic imaging. Tc-99m is best known ...

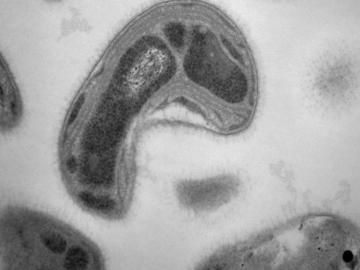
A team led by the Department of Energy’s Oak Ridge National Laboratory has identified a novel microbial process that can break down toxic methylmercury in the environment, a fundamental scientific discovery that could potentially reduce mercury toxicity levels and sup...

An Oak Ridge National Laboratory study is providing an unprecedented watershed-scale understanding of mercury in soils and sediments. Researchers focused on evaluating mercury and soil properties along the banks of a mercury-contaminated stream in Oak Ridge, Tenn., sampling 145 loca...




