Filter News
Area of Research
- (-) Materials (14)
- (-) Nuclear Science and Technology (13)
- Advanced Manufacturing (1)
- Biology and Environment (8)
- Clean Energy (48)
- Computational Biology (1)
- Computational Engineering (1)
- Computer Science (6)
- Electricity and Smart Grid (1)
- Energy Sciences (1)
- Fusion and Fission (9)
- Fusion Energy (7)
- Isotopes (1)
- National Security (7)
- Neutron Science (3)
- Nuclear Systems Modeling, Simulation and Validation (1)
- Quantum information Science (1)
- Sensors and Controls (1)
- Supercomputing (10)
News Type
News Topics
- (-) Energy Storage (8)
- (-) Fusion (5)
- (-) Nuclear Energy (18)
- 3-D Printing/Advanced Manufacturing (6)
- Advanced Reactors (6)
- Bioenergy (2)
- Biomedical (3)
- Buildings (1)
- Chemical Sciences (6)
- Clean Water (1)
- Composites (4)
- Computer Science (1)
- Coronavirus (1)
- Critical Materials (5)
- Decarbonization (1)
- Environment (2)
- Isotopes (3)
- Materials (15)
- Materials Science (25)
- Microscopy (9)
- Molten Salt (4)
- Nanotechnology (12)
- Neutron Science (8)
- Physics (9)
- Polymers (8)
- Quantum Computing (2)
- Quantum Science (1)
- Space Exploration (3)
- Sustainable Energy (3)
- Transportation (6)
Media Contacts

Electric vehicles can drive longer distances if their lithium-ion batteries deliver more energy in a lighter package. A prime weight-loss candidate is the current collector, a component that often adds 10% to the weight of a battery cell without contributing energy.

ORNL scientists found that a small tweak created big performance improvements in a type of solid-state battery, a technology considered vital to broader electric vehicle adoption.

Andrew Ullman, Distinguished Staff Fellow at Oak Ridge National Laboratory, is using chemistry to devise a better battery
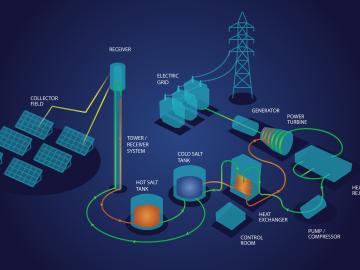
Oak Ridge National Laboratory scientists recently demonstrated a low-temperature, safe route to purifying molten chloride salts that minimizes their ability to corrode metals. This method could make the salts useful for storing energy generated from the sun’s heat.

A developing method to gauge the occurrence of a nuclear reactor anomaly has the potential to save millions of dollars.
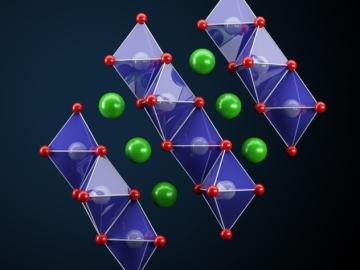
Oak Ridge National Laboratory scientists seeking the source of charge loss in lithium-ion batteries demonstrated that coupling a thin-film cathode with a solid electrolyte is a rapid way to determine the root cause.

In the search to create materials that can withstand extreme radiation, Yanwen Zhang, a researcher at the Department of Energy’s Oak Ridge National Laboratory, says that materials scientists must think outside the box.

Oak Ridge National Laboratory researchers have developed a thin film, highly conductive solid-state electrolyte made of a polymer and ceramic-based composite for lithium metal batteries.

In the 1960s, Oak Ridge National Laboratory's four-year Molten Salt Reactor Experiment tested the viability of liquid fuel reactors for commercial power generation. Results from that historic experiment recently became the basis for the first-ever molten salt reactor benchmark.

As a teenager, Kat Royston had a lot of questions. Then an advanced-placement class in physics convinced her all the answers were out there.




