Filter News
Area of Research
- (-) Neutron Science (26)
- Advanced Manufacturing (1)
- Biology and Environment (30)
- Clean Energy (55)
- Climate and Environmental Systems (1)
- Computational Engineering (1)
- Computer Science (3)
- Fusion and Fission (5)
- Isotope Development and Production (1)
- Isotopes (3)
- Materials (51)
- Materials Characterization (1)
- Materials for Computing (7)
- Materials Under Extremes (1)
- National Security (9)
- Nuclear Science and Technology (3)
- Quantum information Science (1)
- Sensors and Controls (1)
- Supercomputing (32)
News Topics
- (-) Bioenergy (3)
- (-) Biomedical (4)
- (-) Coronavirus (5)
- (-) Environment (3)
- (-) Materials Science (13)
- (-) Quantum Science (4)
- (-) Security (1)
- (-) Transportation (2)
- 3-D Printing/Advanced Manufacturing (3)
- Artificial Intelligence (1)
- Big Data (1)
- Biology (4)
- Biotechnology (1)
- Climate Change (1)
- Composites (1)
- Computer Science (6)
- Cybersecurity (1)
- Decarbonization (1)
- Energy Storage (2)
- Frontier (1)
- Fusion (1)
- High-Performance Computing (1)
- Materials (6)
- Microscopy (1)
- Nanotechnology (6)
- National Security (1)
- Neutron Science (40)
- Nuclear Energy (1)
- Physics (7)
- Space Exploration (1)
- Summit (4)
- Sustainable Energy (2)
Media Contacts
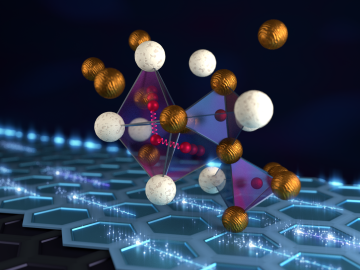
An international team of researchers has discovered the hydrogen atoms in a metal hydride material are much more tightly spaced than had been predicted for decades — a feature that could possibly facilitate superconductivity at or near room temperature and pressure.
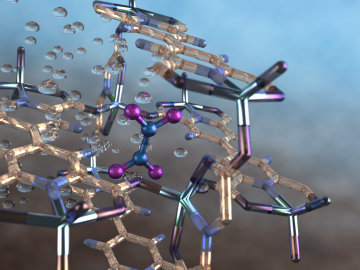
An international team of scientists, led by the University of Manchester, has developed a metal-organic framework, or MOF, material
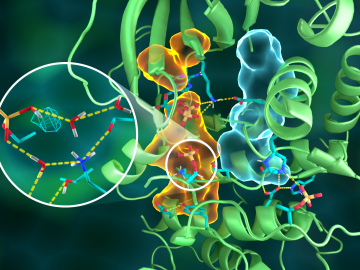
OAK RIDGE, Tenn., March 20, 2019—Direct observations of the structure and catalytic mechanism of a prototypical kinase enzyme—protein kinase A or PKA—will provide researchers and drug developers with significantly enhanced abilities to understand and treat fatal diseases and neurological disorders such as cancer, diabetes, and cystic fibrosis.

Scientists at the Department of Energy’s Oak Ridge National Laboratory have created a recipe for a renewable 3D printing feedstock that could spur a profitable new use for an intractable biorefinery byproduct: lignin.

Researchers used neutrons to probe a running engine at ORNL’s Spallation Neutron Source
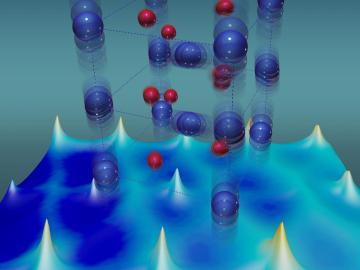
For more than 50 years, scientists have debated what turns particular oxide insulators, in which electrons barely move, into metals, in which electrons flow freely.




