Filter News
Area of Research
- (-) Materials (28)
- Advanced Manufacturing (3)
- Biology and Environment (10)
- Clean Energy (45)
- Computational Engineering (1)
- Computer Science (4)
- Fuel Cycle Science and Technology (1)
- Fusion and Fission (11)
- Fusion Energy (1)
- Isotope Development and Production (1)
- Isotopes (2)
- Materials for Computing (3)
- National Security (15)
- Neutron Science (10)
- Nuclear Science and Technology (7)
- Quantum information Science (1)
- Sensors and Controls (1)
- Supercomputing (45)
News Topics
- (-) Advanced Reactors (1)
- (-) Artificial Intelligence (4)
- (-) Computer Science (8)
- (-) Energy Storage (19)
- (-) Exascale Computing (1)
- (-) Grid (2)
- (-) Machine Learning (2)
- (-) Nuclear Energy (2)
- (-) Security (1)
- 3-D Printing/Advanced Manufacturing (13)
- Bioenergy (8)
- Biology (4)
- Biomedical (3)
- Buildings (2)
- Chemical Sciences (20)
- Climate Change (5)
- Composites (3)
- Coronavirus (2)
- Critical Materials (8)
- Cybersecurity (3)
- Decarbonization (4)
- Environment (7)
- Frontier (2)
- Fusion (2)
- High-Performance Computing (2)
- Isotopes (5)
- ITER (1)
- Materials (38)
- Materials Science (35)
- Microscopy (12)
- Molten Salt (2)
- Nanotechnology (21)
- National Security (3)
- Net Zero (1)
- Neutron Science (18)
- Partnerships (8)
- Physics (14)
- Polymers (6)
- Quantum Computing (1)
- Quantum Science (10)
- Renewable Energy (1)
- Summit (1)
- Sustainable Energy (7)
- Transformational Challenge Reactor (1)
- Transportation (4)
Media Contacts

Momentum Technologies Inc., a Dallas, Texas-based materials science company that is focused on extracting critical metals from electronic waste, has licensed an Oak Ridge National Laboratory process for recovering cobalt and other metals from spent
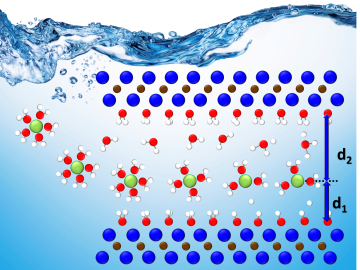
A team led by Oak Ridge National Laboratory developed a novel, integrated approach to track energy-transporting ions within an ultra-thin material, which could unlock its energy storage potential leading toward faster charging, longer-lasting devices.

Five researchers at the Department of Energy’s Oak Ridge National Laboratory have been named ORNL Corporate Fellows in recognition of significant career accomplishments and continued leadership in their scientific fields.

Energy storage startup SPARKZ Inc. has exclusively licensed five battery technologies from the Department of Energy’s Oak Ridge National Laboratory designed to eliminate cobalt metal in lithium-ion batteries. The advancement is aimed at accelerating the production of electric vehicles and energy storage solutions for the power grid.
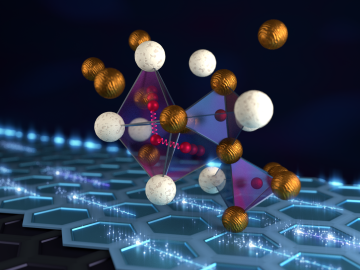
An international team of researchers has discovered the hydrogen atoms in a metal hydride material are much more tightly spaced than had been predicted for decades — a feature that could possibly facilitate superconductivity at or near room temperature and pressure.

Researchers at the Department of Energy’s Oak Ridge National Laboratory have received five 2019 R&D 100 Awards, increasing the lab’s total to 221 since the award’s inception in 1963.
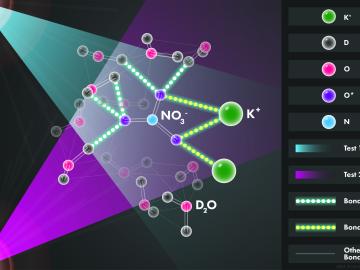
Scientists at the Department of Energy’s Oak Ridge National Laboratory used neutrons, isotopes and simulations to “see” the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come




