Filter News
Area of Research
- (-) Materials for Computing (2)
- (-) Neutron Science (8)
- (-) Nuclear Science and Technology (3)
- Biology and Environment (10)
- Clean Energy (12)
- Computational Engineering (1)
- Fusion and Fission (4)
- Fusion Energy (1)
- Isotopes (2)
- Materials (7)
- National Security (6)
- Sensors and Controls (1)
- Supercomputing (22)
News Topics
- (-) Advanced Reactors (2)
- (-) Biomedical (6)
- (-) Coronavirus (5)
- (-) Frontier (1)
- (-) Security (1)
- 3-D Printing/Advanced Manufacturing (7)
- Artificial Intelligence (1)
- Big Data (1)
- Bioenergy (4)
- Biology (4)
- Biotechnology (1)
- Chemical Sciences (2)
- Climate Change (1)
- Composites (2)
- Computer Science (8)
- Cybersecurity (1)
- Decarbonization (2)
- Energy Storage (4)
- Environment (4)
- Fusion (2)
- High-Performance Computing (1)
- Isotopes (3)
- Materials (8)
- Materials Science (17)
- Microscopy (2)
- Nanotechnology (7)
- National Security (2)
- Neutron Science (40)
- Nuclear Energy (8)
- Physics (7)
- Polymers (4)
- Quantum Science (5)
- Space Exploration (3)
- Summit (4)
- Sustainable Energy (5)
- Transformational Challenge Reactor (1)
- Transportation (3)
Media Contacts
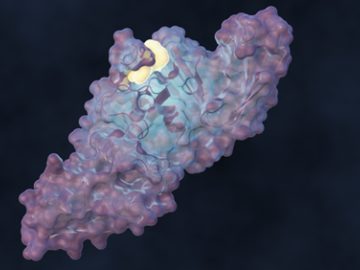
A team of scientists led by the Department of Energy’s Oak Ridge National Laboratory designed a molecule that disrupts the infection mechanism of the SARS-CoV-2 coronavirus and could be used to develop new treatments for COVID-19 and other viral diseases.

Paul Langan will join ORNL in the spring as associate laboratory director for the Biological and Environmental Systems Science Directorate.

More than 50 current employees and recent retirees from ORNL received Department of Energy Secretary’s Honor Awards from Secretary Jennifer Granholm in January as part of project teams spanning the national laboratory system. The annual awards recognized 21 teams and three individuals for service and contributions to DOE’s mission and to the benefit of the nation.
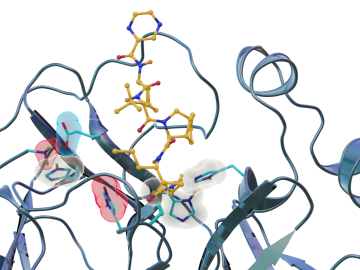
Scientists have found new, unexpected behaviors when SARS-CoV-2 – the virus that causes COVID-19 – encounters drugs known as inhibitors, which bind to certain components of the virus and block its ability to reproduce.
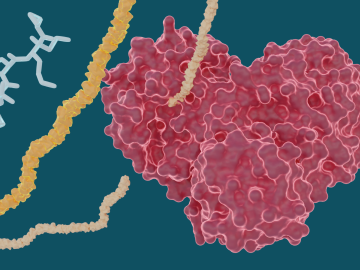
Experiments led by researchers at ORNL have determined that several hepatitis C drugs can inhibit the SARS-CoV-2 main protease, a crucial protein enzyme that enables the novel coronavirus to reproduce.
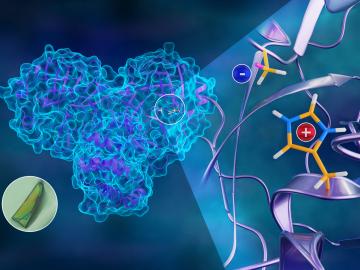
To better understand how the novel coronavirus behaves and how it can be stopped, scientists have completed a three-dimensional map that reveals the location of every atom in an enzyme molecule critical to SARS-CoV-2 reproduction.

Four research teams from the Department of Energy’s Oak Ridge National Laboratory and their technologies have received 2020 R&D 100 Awards.
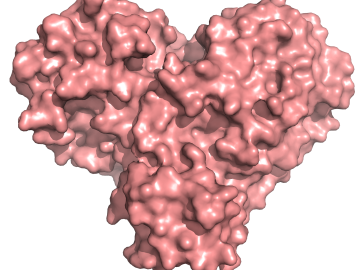
A team of researchers has performed the first room-temperature X-ray measurements on the SARS-CoV-2 main protease — the enzyme that enables the virus to reproduce.

Researchers at the Department of Energy’s Oak Ridge National Laboratory are refining their design of a 3D-printed nuclear reactor core, scaling up the additive manufacturing process necessary to build it, and developing methods
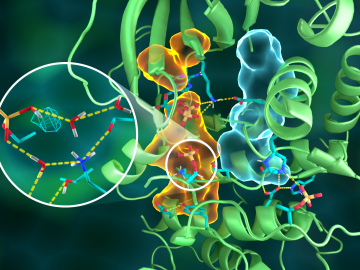
OAK RIDGE, Tenn., March 20, 2019—Direct observations of the structure and catalytic mechanism of a prototypical kinase enzyme—protein kinase A or PKA—will provide researchers and drug developers with significantly enhanced abilities to understand and treat fatal diseases and neurological disorders such as cancer, diabetes, and cystic fibrosis.




