Filter News
Area of Research
News Topics
- (-) Chemical Sciences (7)
- (-) Energy Storage (6)
- 3-D Printing/Advanced Manufacturing (4)
- Advanced Reactors (1)
- Artificial Intelligence (4)
- Big Data (2)
- Bioenergy (2)
- Biomedical (2)
- Buildings (1)
- Clean Water (2)
- Composites (2)
- Computer Science (8)
- Coronavirus (1)
- Cybersecurity (1)
- Decarbonization (1)
- Environment (6)
- Exascale Computing (1)
- Fusion (2)
- Grid (2)
- High-Performance Computing (1)
- Isotopes (6)
- Machine Learning (2)
- Materials (19)
- Materials Science (17)
- Mathematics (1)
- Microscopy (6)
- Nanotechnology (8)
- Neutron Science (9)
- Nuclear Energy (9)
- Partnerships (3)
- Physics (11)
- Polymers (4)
- Quantum Computing (1)
- Security (1)
- Space Exploration (1)
- Summit (1)
- Sustainable Energy (2)
- Transformational Challenge Reactor (2)
- Transportation (4)
Media Contacts

The founder of a startup company who is working with ORNL has won an Environmental Protection Agency Green Chemistry Challenge Award for a unique air pollution control technology.

In response to a renewed international interest in molten salt reactors, researchers from the Department of Energy’s Oak Ridge National Laboratory have developed a novel technique to visualize molten salt intrusion in graphite.

In fiscal year 2023 — Oct. 1–Sept. 30, 2023 — Oak Ridge National Laboratory was awarded more than $8 million in technology maturation funding through the Department of Energy’s Technology Commercialization Fund, or TCF.

Little of the mixed consumer plastics thrown away or placed in recycle bins actually ends up being recycled. Nearly 90% is buried in landfills or incinerated at commercial facilities that generate greenhouse gases and airborne toxins. Neither outcome is ideal for the environment.

Xiao-Ying Yu, a distinguished scientist in the Materials Science and Technology Division of the Department of Energy’s Oak Ridge National Laboratory, has recently been chosen for several prominent editorial roles.
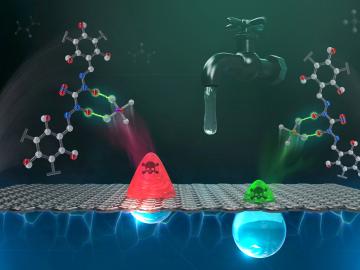
Researchers at ORNL are tackling a global water challenge with a unique material designed to target not one, but two toxic, heavy metal pollutants for simultaneous removal.

At the Department of Energy’s Oak Ridge National Laboratory, scientists use artificial intelligence, or AI, to accelerate the discovery and development of materials for energy and information technologies.
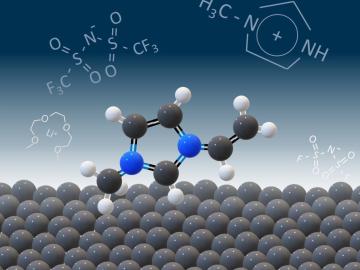
Scientists seeking ways to improve a battery’s ability to hold a charge longer, using advanced materials that are safe, stable and efficient, have determined that the materials themselves are only part of the solution.

The formation of lithium dendrites is still a mystery, but materials engineers study the conditions that enable dendrites and how to stop them.
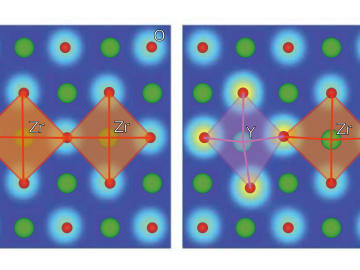
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.




