Filter News
Area of Research
- (-) Neutron Science (13)
- Biological Systems (1)
- Biology and Environment (30)
- Clean Energy (22)
- Computational Biology (1)
- Computational Engineering (1)
- Fusion and Fission (7)
- Fusion Energy (1)
- Isotope Development and Production (1)
- Isotopes (7)
- Materials (23)
- Materials for Computing (6)
- National Security (4)
- Nuclear Science and Technology (10)
- Supercomputing (46)
News Topics
- (-) Biomedical (9)
- (-) Clean Water (2)
- (-) Frontier (1)
- (-) Polymers (1)
- (-) Space Exploration (2)
- 3-D Printing/Advanced Manufacturing (6)
- Artificial Intelligence (5)
- Big Data (2)
- Bioenergy (5)
- Biology (5)
- Biotechnology (1)
- Chemical Sciences (1)
- Climate Change (1)
- Composites (1)
- Computer Science (13)
- Coronavirus (8)
- Cybersecurity (1)
- Decarbonization (2)
- Energy Storage (4)
- Environment (6)
- Fossil Energy (1)
- Fusion (1)
- High-Performance Computing (2)
- Machine Learning (3)
- Materials (11)
- Materials Science (20)
- Mathematics (1)
- Microscopy (2)
- Nanotechnology (8)
- National Security (2)
- Neutron Science (73)
- Nuclear Energy (2)
- Physics (8)
- Quantum Computing (1)
- Quantum Science (5)
- Security (2)
- Summit (6)
- Sustainable Energy (2)
- Transportation (3)
Media Contacts

Biological membranes, such as the “walls” of most types of living cells, primarily consist of a double layer of lipids, or “lipid bilayer,” that forms the structure, and a variety of embedded and attached proteins with highly specialized functions, including proteins that rapidly and selectively transport ions and molecules in and out of the cell.

Researchers at the Department of Energy’s Oak Ridge National Laboratory, Pacific Northwest National Laboratory and Washington State University teamed up to investigate the complex dynamics of low-water liquids that challenge nuclear waste processing at federal cleanup sites.
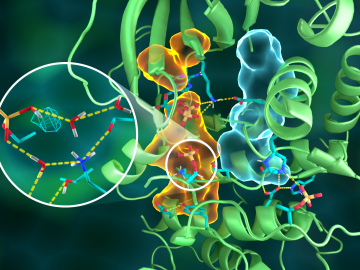
OAK RIDGE, Tenn., March 20, 2019—Direct observations of the structure and catalytic mechanism of a prototypical kinase enzyme—protein kinase A or PKA—will provide researchers and drug developers with significantly enhanced abilities to understand and treat fatal diseases and neurological disorders such as cancer, diabetes, and cystic fibrosis.




