Filter News
Area of Research
News Topics
Media Contacts

Paul Langan will join ORNL in the spring as associate laboratory director for the Biological and Environmental Systems Science Directorate.

Three ORNL scientists have been elected fellows of the American Association for the Advancement of Science, or AAAS, the world’s largest general scientific society and publisher of the Science family of journals.
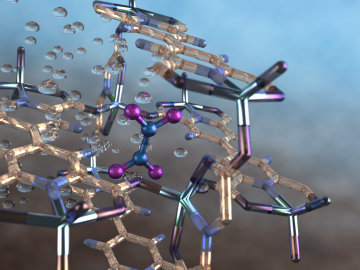
An international team of scientists, led by the University of Manchester, has developed a metal-organic framework, or MOF, material
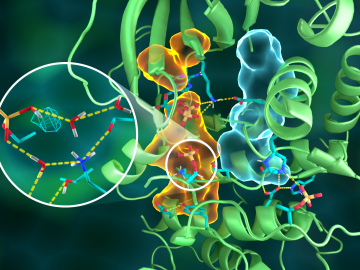
OAK RIDGE, Tenn., March 20, 2019—Direct observations of the structure and catalytic mechanism of a prototypical kinase enzyme—protein kinase A or PKA—will provide researchers and drug developers with significantly enhanced abilities to understand and treat fatal diseases and neurological disorders such as cancer, diabetes, and cystic fibrosis.




