Filter News
Area of Research
News Topics
- (-) Energy Storage (2)
- (-) Machine Learning (2)
- (-) Microscopy (2)
- (-) Summit (1)
- 3-D Printing/Advanced Manufacturing (1)
- Artificial Intelligence (1)
- Big Data (1)
- Bioenergy (3)
- Biomedical (3)
- Clean Water (1)
- Computer Science (4)
- Coronavirus (2)
- Environment (2)
- Materials Science (2)
- Mathematics (1)
- Nanotechnology (1)
- Neutron Science (10)
- Nuclear Energy (1)
- Polymers (1)
- Quantum Science (2)
- Security (1)
- Sustainable Energy (1)
Media Contacts
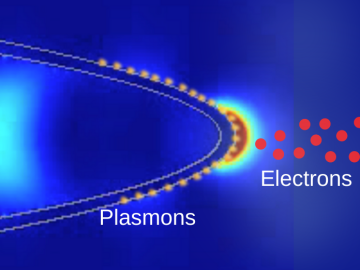
Scientists at ORNL and the University of Nebraska have developed an easier way to generate electrons for nanoscale imaging and sensing, providing a useful new tool for material science, bioimaging and fundamental quantum research.
In the race to identify solutions to the COVID-19 pandemic, researchers at the Department of Energy’s Oak Ridge National Laboratory are joining the fight by applying expertise in computational science, advanced manufacturing, data science and neutron science.
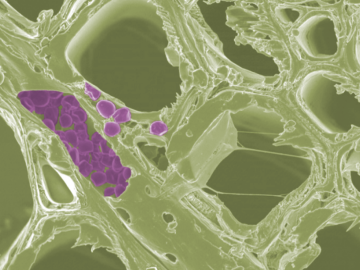
Scientists at the Department of Energy’s Oak Ridge National Laboratory have developed a new method to peer deep into the nanostructure of biomaterials without damaging the sample. This novel technique can confirm structural features in starch, a carbohydrate important in biofuel production.

ORNL computer scientist Catherine Schuman returned to her alma mater, Harriman High School, to lead Hour of Code activities and talk to students about her job as a researcher.

Two of the researchers who share the Nobel Prize in Chemistry announced Wednesday—John B. Goodenough of the University of Texas at Austin and M. Stanley Whittingham of Binghamton University in New York—have research ties to ORNL.
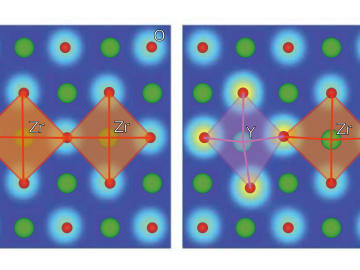
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.




