Filter News
Area of Research
- (-) Biological Systems (1)
- (-) Climate and Environmental Systems (1)
- (-) Materials (14)
- (-) Neutron Science (6)
- (-) Transportation Systems (1)
- Advanced Manufacturing (3)
- Clean Energy (15)
- Computational Engineering (1)
- Computer Science (7)
- Fusion Energy (2)
- National Security (2)
- Quantum information Science (2)
- Supercomputing (16)
News Type
News Topics
- (-) Bioenergy (2)
- (-) Biomedical (2)
- (-) Computer Science (4)
- (-) Materials Science (9)
- (-) Molten Salt (1)
- (-) Nanotechnology (3)
- (-) Sustainable Energy (3)
- 3-D Printing/Advanced Manufacturing (3)
- Advanced Reactors (1)
- Artificial Intelligence (1)
- Clean Water (2)
- Energy Storage (5)
- Environment (7)
- Fusion (2)
- Machine Learning (1)
- Microscopy (2)
- Neutron Science (12)
- Nuclear Energy (5)
- Physics (1)
- Polymers (1)
- Quantum Science (1)
- Transportation (4)
Media Contacts
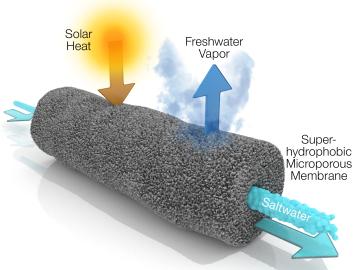
A new method developed at Oak Ridge National Laboratory improves the energy efficiency of a desalination process known as solar-thermal evaporation.
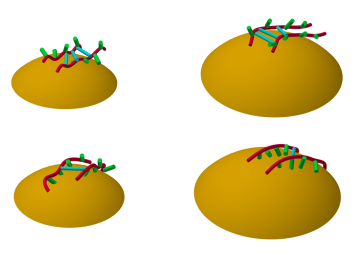
A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.

Oak Ridge National Laboratory has teamed with Cornell College and the University of Tennessee to study ways to repurpose waste soft drinks for carbon capture that could help cut carbon dioxide emissions.
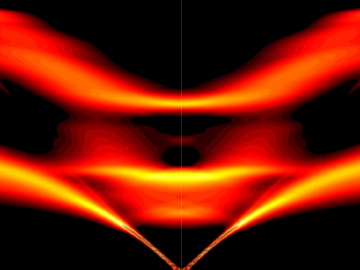
Scientists have discovered a way to alter heat transport in thermoelectric materials, a finding that may ultimately improve energy efficiency as the materials
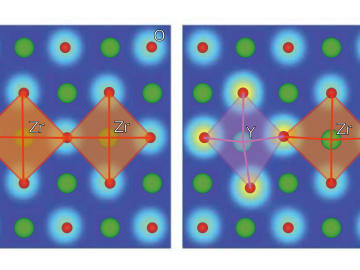
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.
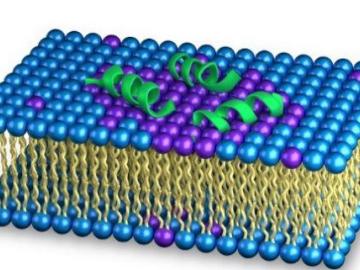
As the rise of antibiotic-resistant bacteria known as superbugs threatens public health, Oak Ridge National Laboratory’s Shuo Qian and Veerendra Sharma from the Bhaba Atomic Research Centre in India are using neutron scattering to study how an antibacterial peptide interacts with and fights harmful bacteria.

Scientists have tested a novel heat-shielding graphite foam, originally created at Oak Ridge National Laboratory, at Germany’s Wendelstein 7-X stellarator with promising results for use in plasma-facing components of fusion reactors.

Oak Ridge National Laboratory scientists analyzed more than 50 years of data showing puzzlingly inconsistent trends about corrosion of structural alloys in molten salts and found one factor mattered most—salt purity.
![Coexpression_hi-res_image[1].jpg Coexpression_hi-res_image[1].jpg](/sites/default/files/styles/list_page_thumbnail/public/Coexpression_hi-res_image%5B1%5D_0.jpg?itok=OnLe-krT)
While studying the genes in poplar trees that control callus formation, scientists at Oak Ridge National Laboratory have uncovered genetic networks at the root of tumor formation in several human cancers.
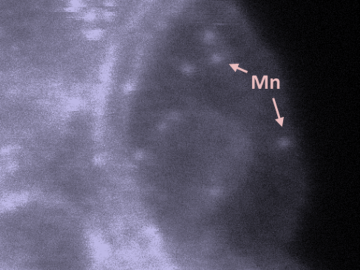
Oak Ridge National Laboratory scientists studying fuel cells as a potential alternative to internal combustion engines used sophisticated electron microscopy to investigate the benefits of replacing high-cost platinum with a lower cost, carbon-nitrogen-manganese-based catalyst.




