Filter News
Area of Research
- (-) Biology and Environment (3)
- (-) National Security (3)
- (-) Neutron Science (6)
- Advanced Manufacturing (1)
- Clean Energy (10)
- Computational Engineering (1)
- Computer Science (2)
- Fusion and Fission (1)
- Isotopes (1)
- Materials (12)
- Materials for Computing (2)
- Quantum information Science (1)
- Supercomputing (16)
News Topics
- (-) Artificial Intelligence (1)
- (-) Bioenergy (2)
- (-) Biomedical (2)
- (-) Computer Science (3)
- (-) Machine Learning (1)
- (-) Materials Science (3)
- (-) Security (1)
- 3-D Printing/Advanced Manufacturing (1)
- Biology (4)
- Biotechnology (1)
- Chemical Sciences (1)
- Climate Change (1)
- Composites (1)
- Coronavirus (1)
- Cybersecurity (2)
- Environment (3)
- Fusion (1)
- Materials (2)
- Microscopy (1)
- Nanotechnology (2)
- National Security (1)
- Neutron Science (12)
- Physics (2)
- Polymers (1)
- Quantum Science (1)
- Sustainable Energy (1)
Media Contacts
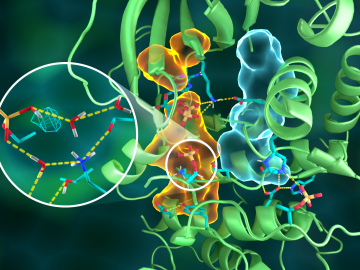
OAK RIDGE, Tenn., March 20, 2019—Direct observations of the structure and catalytic mechanism of a prototypical kinase enzyme—protein kinase A or PKA—will provide researchers and drug developers with significantly enhanced abilities to understand and treat fatal diseases and neurological disorders such as cancer, diabetes, and cystic fibrosis.




