Filter News
Area of Research
- (-) Materials (21)
- Advanced Manufacturing (2)
- Biological Systems (1)
- Biology and Environment (2)
- Clean Energy (16)
- Climate and Environmental Systems (1)
- Computational Engineering (1)
- Computer Science (3)
- National Security (1)
- Neutron Science (6)
- Nuclear Science and Technology (4)
- Supercomputing (18)
News Topics
- (-) Bioenergy (3)
- (-) Biomedical (3)
- (-) Composites (3)
- (-) Microscopy (9)
- (-) Molten Salt (1)
- (-) Polymers (5)
- 3-D Printing/Advanced Manufacturing (7)
- Advanced Reactors (1)
- Artificial Intelligence (1)
- Clean Water (2)
- Computer Science (4)
- Cybersecurity (1)
- Energy Storage (5)
- Environment (5)
- Fusion (3)
- Grid (1)
- Isotopes (6)
- Materials Science (25)
- Nanotechnology (12)
- Neutron Science (7)
- Nuclear Energy (8)
- Physics (6)
- Quantum Science (2)
- Space Exploration (1)
- Sustainable Energy (5)
- Transportation (6)
Media Contacts

Researchers at the Department of Energy’s Oak Ridge National Laboratory have received five 2019 R&D 100 Awards, increasing the lab’s total to 221 since the award’s inception in 1963.

ORNL and The University of Toledo have entered into a memorandum of understanding for collaborative research.
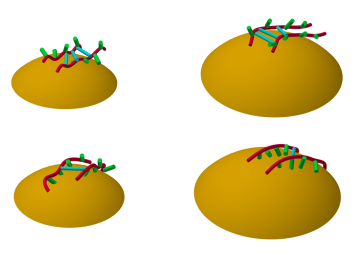
A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.
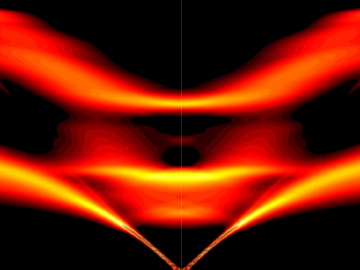
Scientists have discovered a way to alter heat transport in thermoelectric materials, a finding that may ultimately improve energy efficiency as the materials

OAK RIDGE, Tenn., March 22, 2019 – Karren Leslie More, a researcher at the Department of Energy’s Oak Ridge National Laboratory, has been elected fellow of the Microscopy Society of America (MSA) professional organization.
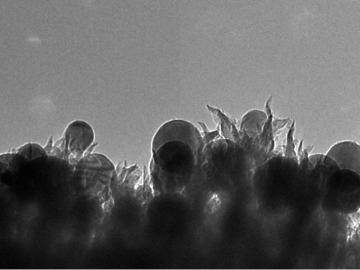
OAK RIDGE, Tenn., March 1, 2019—ReactWell, LLC, has licensed a novel waste-to-fuel technology from the Department of Energy’s Oak Ridge National Laboratory to improve energy conversion methods for cleaner, more efficient oil and gas, chemical and

Vera Bocharova at the Department of Energy’s Oak Ridge National Laboratory investigates the structure and dynamics of soft materials—polymer nanocomposites, polymer electrolytes and biological macromolecules—to advance materials and technologies for energy, medicine and other applications.

Oak Ridge National Laboratory scientists analyzed more than 50 years of data showing puzzlingly inconsistent trends about corrosion of structural alloys in molten salts and found one factor mattered most—salt purity.

OAK RIDGE, Tenn., Jan. 31, 2019—A new electron microscopy technique that detects the subtle changes in the weight of proteins at the nanoscale—while keeping the sample intact—could open a new pathway for deeper, more comprehensive studies of the basic building blocks of life.
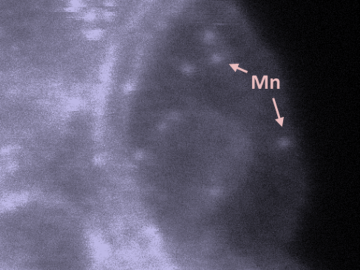
Oak Ridge National Laboratory scientists studying fuel cells as a potential alternative to internal combustion engines used sophisticated electron microscopy to investigate the benefits of replacing high-cost platinum with a lower cost, carbon-nitrogen-manganese-based catalyst.




