Filter News
Area of Research
- (-) Materials (24)
- Biology and Environment (11)
- Clean Energy (31)
- Climate and Environmental Systems (2)
- Computer Science (2)
- Energy Sciences (1)
- Fusion Energy (1)
- Isotopes (4)
- Materials for Computing (4)
- National Security (5)
- Neutron Science (10)
- Nuclear Science and Technology (6)
- Quantum information Science (3)
- Supercomputing (22)
News Topics
- (-) Biomedical (4)
- (-) Climate Change (1)
- (-) Critical Materials (2)
- (-) Cybersecurity (1)
- (-) Energy Storage (9)
- (-) Isotopes (6)
- (-) Quantum Science (4)
- (-) Space Exploration (1)
- 3-D Printing/Advanced Manufacturing (6)
- Advanced Reactors (1)
- Artificial Intelligence (2)
- Big Data (2)
- Bioenergy (4)
- Chemical Sciences (2)
- Composites (2)
- Computer Science (9)
- Coronavirus (1)
- Environment (3)
- Exascale Computing (1)
- Fusion (1)
- Grid (1)
- Machine Learning (3)
- Materials (1)
- Materials Science (37)
- Mathematics (1)
- Microscopy (9)
- Molten Salt (1)
- Nanotechnology (18)
- National Security (1)
- Neutron Science (13)
- Nuclear Energy (5)
- Physics (12)
- Polymers (7)
- Security (1)
- Summit (2)
- Sustainable Energy (5)
- Transformational Challenge Reactor (2)
- Transportation (4)
Media Contacts
Oak Ridge National Laboratory scientists seeking the source of charge loss in lithium-ion batteries demonstrated that coupling a thin-film cathode with a solid electrolyte is a rapid way to determine the root cause.
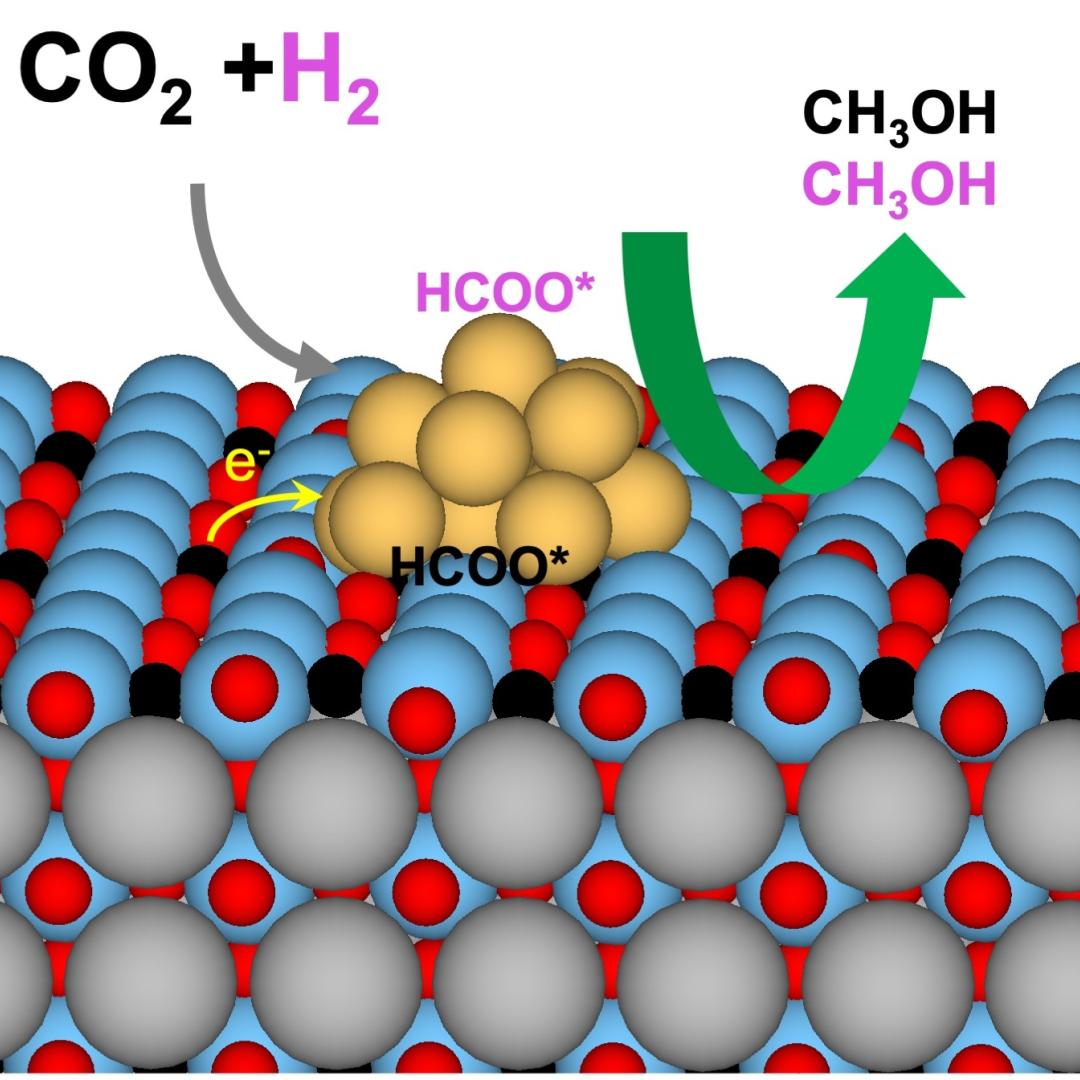
A team led by the Department of Energy’s Oak Ridge National Laboratory synthesized a tiny structure with high surface area and discovered how its unique architecture drives ions across interfaces to transport energy or information.

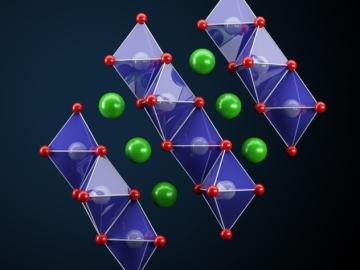
Oak Ridge National Laboratory researchers have developed a thin film, highly conductive solid-state electrolyte made of a polymer and ceramic-based composite for lithium metal batteries.
In the race to identify solutions to the COVID-19 pandemic, researchers at the Department of Energy’s Oak Ridge National Laboratory are joining the fight by applying expertise in computational science, advanced manufacturing, data science and neutron science.

Energy storage startup SPARKZ Inc. has exclusively licensed five battery technologies from the Department of Energy’s Oak Ridge National Laboratory designed to eliminate cobalt metal in lithium-ion batteries. The advancement is aimed at accelerating the production of electric vehicles and energy storage solutions for the power grid.

The formation of lithium dendrites is still a mystery, but materials engineers study the conditions that enable dendrites and how to stop them.
Physicists turned to the “doubly magic” tin isotope Sn-132, colliding it with a target at Oak Ridge National Laboratory to assess its properties as it lost a neutron to become Sn-131.
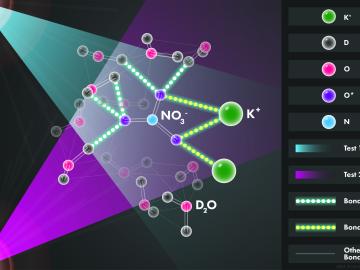
Scientists at the Department of Energy’s Oak Ridge National Laboratory used neutrons, isotopes and simulations to “see” the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come

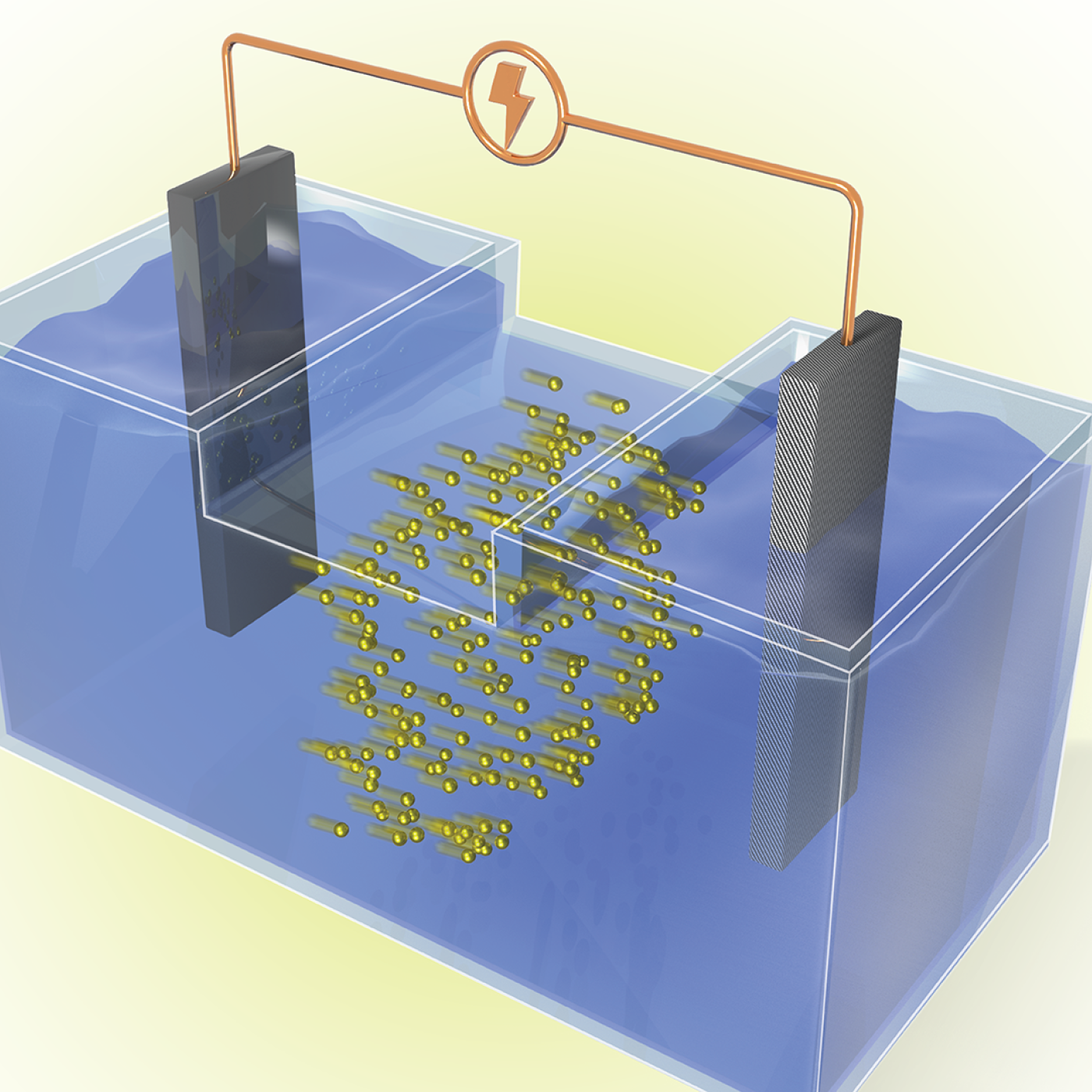
Oak Ridge National Laboratory scientists have developed a crucial component for a new kind of low-cost stationary battery system utilizing common materials and designed for grid-scale electricity storage. Large, economical electricity storage systems can benefit the nation’s grid ...

A tiny vial of gray powder produced at the Department of Energy’s Oak Ridge National Laboratory is the backbone of a new experiment to study the intense magnetic fields created in nuclear collisions.