Filter News
Area of Research
- (-) Materials (11)
- (-) National Security (3)
- Advanced Manufacturing (1)
- Biological Systems (1)
- Biology and Environment (1)
- Clean Energy (17)
- Climate and Environmental Systems (1)
- Computational Engineering (1)
- Computer Science (8)
- Neutron Science (20)
- Quantum information Science (3)
- Supercomputing (26)
News Topics
- (-) Bioenergy (2)
- (-) Computer Science (5)
- (-) Molten Salt (1)
- (-) Neutron Science (4)
- (-) Polymers (2)
- (-) Security (1)
- 3-D Printing/Advanced Manufacturing (6)
- Advanced Reactors (1)
- Artificial Intelligence (2)
- Big Data (1)
- Clean Water (2)
- Composites (1)
- Critical Materials (1)
- Cybersecurity (2)
- Energy Storage (4)
- Environment (5)
- Fusion (2)
- Grid (1)
- Isotopes (1)
- Machine Learning (1)
- Materials Science (18)
- Microscopy (5)
- Nanotechnology (6)
- Nuclear Energy (7)
- Physics (2)
- Quantum Science (2)
- Sustainable Energy (5)
- Transportation (5)
Media Contacts
OAK RIDGE, Tenn., March 1, 2019—ReactWell, LLC, has licensed a novel waste-to-fuel technology from the Department of Energy’s Oak Ridge National Laboratory to improve energy conversion methods for cleaner, more efficient oil and gas, chemical and

Vera Bocharova at the Department of Energy’s Oak Ridge National Laboratory investigates the structure and dynamics of soft materials—polymer nanocomposites, polymer electrolytes and biological macromolecules—to advance materials and technologies for energy, medicine and other applications.
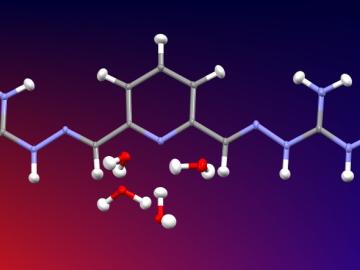
Researchers used neutron scattering at Oak Ridge National Laboratory’s Spallation Neutron Source to investigate the effectiveness of a novel crystallization method to capture carbon dioxide directly from the air.

Oak Ridge National Laboratory scientists analyzed more than 50 years of data showing puzzlingly inconsistent trends about corrosion of structural alloys in molten salts and found one factor mattered most—salt purity.




