Filter News
Area of Research
- (-) Clean Energy (13)
- (-) National Security (7)
- (-) Neutron Science (3)
- Advanced Manufacturing (1)
- Biology and Environment (6)
- Computer Science (1)
- Fusion and Fission (1)
- Isotope Development and Production (1)
- Materials (15)
- Materials Characterization (1)
- Materials Under Extremes (1)
- Nuclear Science and Technology (1)
- Supercomputing (10)
News Topics
- (-) Artificial Intelligence (5)
- (-) Bioenergy (6)
- (-) Biomedical (2)
- (-) Materials Science (5)
- (-) Transportation (7)
- 3-D Printing/Advanced Manufacturing (10)
- Advanced Reactors (1)
- Big Data (1)
- Biology (1)
- Biotechnology (2)
- Buildings (2)
- Chemical Sciences (3)
- Clean Water (1)
- Climate Change (2)
- Composites (2)
- Computer Science (4)
- Coronavirus (2)
- Cybersecurity (5)
- Decarbonization (5)
- Energy Storage (4)
- Environment (4)
- Fusion (1)
- Grid (2)
- High-Performance Computing (2)
- Machine Learning (4)
- Materials (6)
- Microscopy (1)
- Nanotechnology (1)
- National Security (3)
- Net Zero (1)
- Neutron Science (11)
- Nuclear Energy (2)
- Partnerships (7)
- Physics (1)
- Renewable Energy (1)
- Security (3)
- Simulation (1)
- Sustainable Energy (6)
Media Contacts
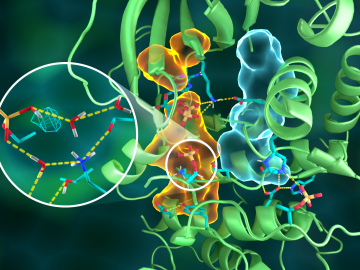
OAK RIDGE, Tenn., March 20, 2019—Direct observations of the structure and catalytic mechanism of a prototypical kinase enzyme—protein kinase A or PKA—will provide researchers and drug developers with significantly enhanced abilities to understand and treat fatal diseases and neurological disorders such as cancer, diabetes, and cystic fibrosis.
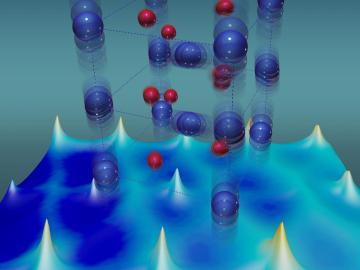
For more than 50 years, scientists have debated what turns particular oxide insulators, in which electrons barely move, into metals, in which electrons flow freely.




