Filter News
Area of Research
- (-) Materials (35)
- (-) Materials for Computing (1)
- (-) Nuclear Science and Technology (7)
- Advanced Manufacturing (4)
- Biological Systems (4)
- Biology and Soft Matter (1)
- Building Technologies (1)
- Chemistry and Physics at Interfaces (1)
- Clean Energy (25)
- Climate and Environmental Systems (1)
- Computational Biology (1)
- Energy Frontier Research Centers (2)
- Fossil Energy (1)
- Functional Materials for Energy (3)
- Geographic Information Science and Technology (1)
- Materials Synthesis from Atoms to Systems (3)
- Materials Under Extremes (1)
- Neutron Science (11)
- Nuclear Systems Technology (1)
- Sensors and Controls (1)
- Supercomputing (12)
News Type
Media Contacts

A collection of seven technologies for lithium recovery developed by scientists from ORNL has been licensed to Element3, a Texas-based company focused on extracting lithium from wastewater produced by oil and gas production.

Electric vehicles can drive longer distances if their lithium-ion batteries deliver more energy in a lighter package. A prime weight-loss candidate is the current collector, a component that often adds 10% to the weight of a battery cell without contributing energy.

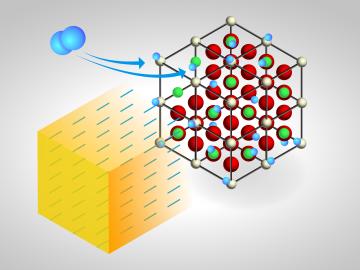
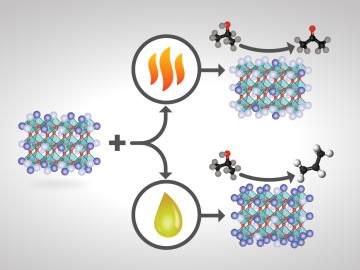
For some crystalline catalysts, what you see on the surface is not always what you get in the bulk, according to two studies led by the Department of Energy’s Oak Ridge National Laboratory. The investigators discovered that treating a complex

New method to detect spin current in quantum materials unlocks potential for alternative electronics

After more than a year of operation at the Department of Energy’s (DOE’s) Oak Ridge National Laboratory (ORNL), the COHERENT experiment, using the world’s smallest neutrino detector, has found a big fingerprint of the elusive, electrically neutral particles that interact only weakly with matter.







