Filter News
Area of Research
- (-) Materials (57)
- (-) Neutron Science (22)
- (-) Quantum information Science (1)
- Advanced Manufacturing (1)
- Biology and Environment (25)
- Clean Energy (46)
- Climate and Environmental Systems (1)
- Computational Engineering (1)
- Computer Science (2)
- Fusion and Fission (4)
- Isotope Development and Production (1)
- Isotopes (7)
- Materials Characterization (1)
- Materials for Computing (7)
- Materials Under Extremes (1)
- National Security (12)
- Nuclear Science and Technology (3)
- Supercomputing (33)
News Topics
- (-) Biomedical (7)
- (-) Cybersecurity (5)
- (-) Energy Storage (20)
- (-) Environment (10)
- (-) Frontier (3)
- (-) Isotopes (5)
- (-) Materials Science (39)
- (-) Space Exploration (1)
- 3-D Printing/Advanced Manufacturing (15)
- Advanced Reactors (1)
- Artificial Intelligence (4)
- Big Data (1)
- Bioenergy (9)
- Biology (8)
- Biotechnology (1)
- Buildings (2)
- Chemical Sciences (19)
- Climate Change (5)
- Composites (3)
- Computer Science (12)
- Coronavirus (7)
- Critical Materials (7)
- Decarbonization (5)
- Exascale Computing (1)
- Fusion (3)
- Grid (2)
- High-Performance Computing (3)
- ITER (1)
- Machine Learning (2)
- Materials (40)
- Microscopy (12)
- Molten Salt (2)
- Nanotechnology (22)
- National Security (3)
- Net Zero (1)
- Neutron Science (42)
- Nuclear Energy (3)
- Partnerships (7)
- Physics (16)
- Polymers (6)
- Quantum Computing (1)
- Quantum Science (12)
- Renewable Energy (1)
- Security (2)
- Summit (4)
- Sustainable Energy (8)
- Transformational Challenge Reactor (1)
- Transportation (6)
Media Contacts

A team led by scientists at the Department of Energy’s Oak Ridge National Laboratory explored how atomically thin two-dimensional (2D) crystals can grow over 3D objects and how the curvature of those objects can stretch and strain the

OAK RIDGE, Tenn., May 7, 2019—Energy Secretary Rick Perry, Congressman Chuck Fleischmann and lab officials today broke ground on a multipurpose research facility that will provide state-of-the-art laboratory space
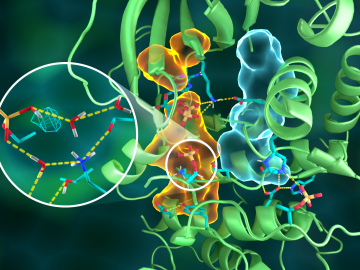
OAK RIDGE, Tenn., March 20, 2019—Direct observations of the structure and catalytic mechanism of a prototypical kinase enzyme—protein kinase A or PKA—will provide researchers and drug developers with significantly enhanced abilities to understand and treat fatal diseases and neurological disorders such as cancer, diabetes, and cystic fibrosis.
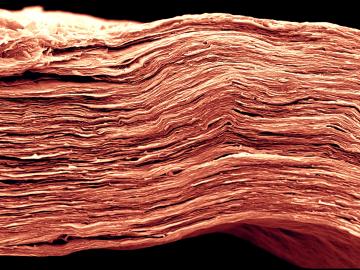
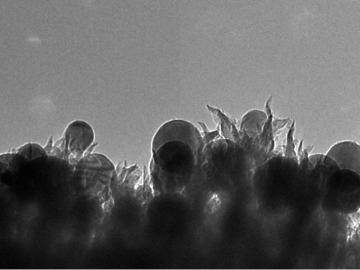
OAK RIDGE, Tenn., March 1, 2019—ReactWell, LLC, has licensed a novel waste-to-fuel technology from the Department of Energy’s Oak Ridge National Laboratory to improve energy conversion methods for cleaner, more efficient oil and gas, chemical and

OAK RIDGE, Tenn., Feb. 8, 2019—The Department of Energy’s Oak Ridge National Laboratory has named Sean Hearne director of the Center for Nanophase Materials Sciences. The center is a DOE Office of Science User Facility that brings world-leading resources and capabilities to the nanoscience resear...

OAK RIDGE, Tenn., Jan. 31, 2019—A new electron microscopy technique that detects the subtle changes in the weight of proteins at the nanoscale—while keeping the sample intact—could open a new pathway for deeper, more comprehensive studies of the basic building blocks of life.
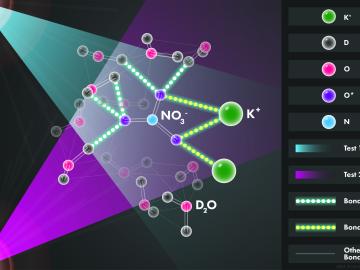
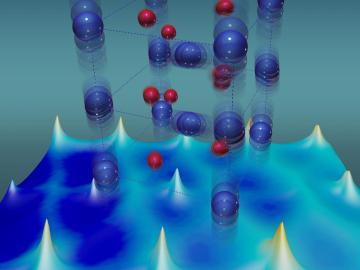
Scientists at the Department of Energy’s Oak Ridge National Laboratory used neutrons, isotopes and simulations to “see” the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come

The Department of Energy’s Oak Ridge National Laboratory is now producing actinium-227 (Ac-227) to meet projected demand for a highly effective cancer drug through a 10-year contract between the U.S. DOE Isotope Program and Bayer.
For more than 50 years, scientists have debated what turns particular oxide insulators, in which electrons barely move, into metals, in which electrons flow freely.




