
Filter News
Area of Research
- (-) Materials (34)
- (-) Neutron Science (7)
- Advanced Manufacturing (1)
- Biology and Environment (6)
- Clean Energy (50)
- Computer Science (2)
- Energy Sciences (1)
- Fusion and Fission (3)
- Fusion Energy (1)
- Isotope Development and Production (1)
- Isotopes (11)
- Materials for Computing (4)
- National Security (3)
- Nuclear Science and Technology (6)
- Supercomputing (20)
News Type
News Topics
- (-) Energy Storage (29)
- (-) Frontier (3)
- (-) Isotopes (7)
- (-) Space Exploration (3)
- 3-D Printing/Advanced Manufacturing (21)
- Advanced Reactors (2)
- Artificial Intelligence (5)
- Big Data (1)
- Bioenergy (11)
- Biology (8)
- Biomedical (11)
- Biotechnology (1)
- Buildings (3)
- Chemical Sciences (25)
- Clean Water (1)
- Climate Change (5)
- Composites (7)
- Computer Science (16)
- Coronavirus (8)
- Critical Materials (13)
- Cybersecurity (6)
- Decarbonization (6)
- Environment (11)
- Exascale Computing (1)
- Fusion (5)
- Grid (3)
- High-Performance Computing (3)
- ITER (1)
- Machine Learning (2)
- Materials (54)
- Materials Science (59)
- Microscopy (18)
- Molten Salt (3)
- Nanotechnology (31)
- National Security (3)
- Net Zero (1)
- Neutron Science (65)
- Nuclear Energy (7)
- Partnerships (8)
- Physics (19)
- Polymers (12)
- Quantum Computing (2)
- Quantum Science (17)
- Renewable Energy (1)
- Security (2)
- Summit (4)
- Sustainable Energy (11)
- Transformational Challenge Reactor (1)
- Transportation (13)
Media Contacts

Electric vehicles can drive longer distances if their lithium-ion batteries deliver more energy in a lighter package. A prime weight-loss candidate is the current collector, a component that often adds 10% to the weight of a battery cell without contributing energy.
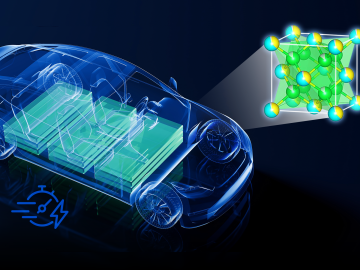
Currently, the biggest hurdle for electric vehicles, or EVs, is the development of advanced battery technology to extend driving range, safety and reliability.

As current courses through a battery, its materials erode over time. Mechanical influences such as stress and strain affect this trajectory, although their impacts on battery efficacy and longevity are not fully understood.

ORNL has been selected to lead an Energy Earthshot Research Center, or EERC, focused on developing chemical processes that use sustainable methods instead of burning fossil fuels to radically reduce industrial greenhouse gas emissions to stem climate change and limit the crisis of a rapidly warming planet.

ORNL scientists found that a small tweak created big performance improvements in a type of solid-state battery, a technology considered vital to broader electric vehicle adoption.

Three scientists from the Department of Energy’s Oak Ridge National Laboratory have been elected fellows of the American Association for the Advancement of Science, or AAAS.

Paul Langan will join ORNL in the spring as associate laboratory director for the Biological and Environmental Systems Science Directorate.

Seven scientists at the Department of Energy’s Oak Ridge National Laboratory have been named Battelle Distinguished Inventors, in recognition of their obtaining 14 or more patents during their careers at the lab.

Three researchers at ORNL have been named ORNL Corporate Fellows in recognition of significant career accomplishments and continued leadership in their scientific fields.

Researchers at ORNL have developed a new method for producing a key component of lithium-ion batteries. The result is a more affordable battery from a faster, less wasteful process that uses less toxic material.


