
Filter News
Area of Research
- Biology and Environment (16)
- Biology and Soft Matter (1)
- Clean Energy (16)
- Computer Science (1)
- Electricity and Smart Grid (1)
- Functional Materials for Energy (1)
- Fusion and Fission (5)
- Fusion Energy (1)
- Materials (35)
- Materials for Computing (4)
- National Security (2)
- Neutron Science (4)
- Supercomputing (43)
News Topics
- (-) Chemical Sciences (74)
- (-) Exascale Computing (44)
- (-) Frontier (46)
- 3-D Printing/Advanced Manufacturing (128)
- Advanced Reactors (35)
- Artificial Intelligence (102)
- Big Data (62)
- Bioenergy (92)
- Biology (102)
- Biomedical (62)
- Biotechnology (24)
- Buildings (67)
- Clean Water (31)
- Climate Change (106)
- Composites (30)
- Computer Science (199)
- Coronavirus (46)
- Critical Materials (29)
- Cybersecurity (35)
- Decarbonization (85)
- Education (5)
- Element Discovery (1)
- Emergency (2)
- Energy Storage (112)
- Environment (201)
- Fossil Energy (6)
- Fusion (59)
- Grid (67)
- High-Performance Computing (94)
- Hydropower (11)
- Irradiation (3)
- Isotopes (57)
- ITER (7)
- Machine Learning (51)
- Materials (150)
- Materials Science (149)
- Mathematics (10)
- Mercury (12)
- Microelectronics (4)
- Microscopy (51)
- Molten Salt (9)
- Nanotechnology (60)
- National Security (73)
- Net Zero (14)
- Neutron Science (140)
- Nuclear Energy (111)
- Partnerships (51)
- Physics (64)
- Polymers (33)
- Quantum Computing (39)
- Quantum Science (73)
- Renewable Energy (2)
- Security (26)
- Simulation (53)
- Software (1)
- Space Exploration (25)
- Statistics (3)
- Summit (61)
- Sustainable Energy (130)
- Transformational Challenge Reactor (7)
- Transportation (99)
Media Contacts

Researchers at ORNL have demonstrated that small molecular tweaks to surfaces can improve absorption technology for direct air capture of carbon dioxide. The team added a charged polymer layer to an amino acid solution, and then, through spectroscopy and simulation, found that the charged layer can hold amino acids at its surface.

Benjamin Manard, an analytical chemist in the Chemical Sciences Division of the Department of Energy’s Oak Ridge National Laboratory, will receive the 2024 Lester W. Strock Award from the Society of Applied Spectroscopy.

Five researchers at the Department of Energy’s Oak Ridge National Laboratory recently completed an eight-week pilot commercialization coaching program as part of Safari, a program funded by DOE’s Office of Technology Transitions, or OTT, Practices to Accelerate the Commercialization of Technologies, or PACT.

The world’s fastest supercomputer helped researchers simulate synthesizing a material harder and tougher than a diamond — or any other substance on Earth. The study used Frontier to predict the likeliest strategy to synthesize such a material, thought to exist so far only within the interiors of giant exoplanets, or planets beyond our solar system.

A team of federal contractor and national laboratory engineers and scientists from the U.S. Department of Energy Office of Environmental Management has been nationally distinguished as “Heroes of Chemistry” for making the world better through their effort, ingenuity, creativity and perseverance.
The contract will be awarded to develop the newest high-performance computing system at the Oak Ridge Leadership Computing Facility.

Researchers conduct largest, most accurate molecular dynamics simulations to date of two million correlated electrons using Frontier, the world’s fastest supercomputer. The simulation, which exceed an exaflop using full double precision, is 1,000 times greater in size and speed than any quantum chemistry simulation of it's kind.

ORNL's Guang Yang and Andrew Westover have been selected to join the first cohort of DOE’s Advanced Research Projects Agency-Energy Inspiring Generations of New Innovators to Impact Technologies in Energy 2024 program. The program supports early career scientists and engineers in their work to convert disruptive ideas into impactful energy technologies.

Oak Ridge National Laboratory scientists have developed a method leveraging artificial intelligence to accelerate the identification of environmentally friendly solvents for industrial carbon capture, biomass processing, rechargeable batteries and other applications.
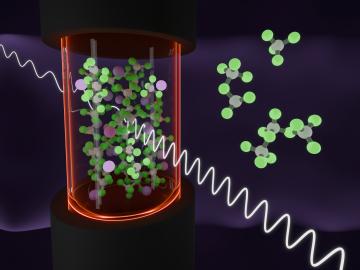
An Oak Ridge National Laboratory team revealed how chemical species form in a highly reactive molten salt mixture of aluminum chloride and potassium chloride by unraveling vibrational signatures and observing ion exchanges.


