Filter News
Area of Research
- (-) Neutron Science (38)
- Advanced Manufacturing (6)
- Biology and Environment (117)
- Biology and Soft Matter (1)
- Clean Energy (116)
- Climate and Environmental Systems (6)
- Computational Biology (1)
- Computational Engineering (2)
- Computer Science (3)
- Electricity and Smart Grid (1)
- Fuel Cycle Science and Technology (1)
- Functional Materials for Energy (1)
- Fusion and Fission (46)
- Fusion Energy (15)
- Isotope Development and Production (1)
- Isotopes (26)
- Materials (121)
- Materials Characterization (1)
- Materials for Computing (20)
- Materials Under Extremes (1)
- Mathematics (1)
- National Security (18)
- Nuclear Science and Technology (40)
- Nuclear Systems Modeling, Simulation and Validation (1)
- Supercomputing (87)
- Transportation Systems (2)
News Topics
- (-) Climate Change (1)
- (-) Decarbonization (2)
- (-) Environment (8)
- (-) Fusion (1)
- (-) Materials Science (23)
- (-) Nuclear Energy (3)
- (-) Polymers (1)
- (-) Summit (6)
- 3-D Printing/Advanced Manufacturing (6)
- Advanced Reactors (1)
- Artificial Intelligence (6)
- Big Data (2)
- Bioenergy (6)
- Biology (5)
- Biomedical (11)
- Biotechnology (1)
- Chemical Sciences (2)
- Clean Water (2)
- Composites (1)
- Computer Science (13)
- Coronavirus (8)
- Cybersecurity (1)
- Energy Storage (6)
- Fossil Energy (1)
- Frontier (1)
- High-Performance Computing (2)
- Machine Learning (3)
- Materials (14)
- Mathematics (1)
- Microscopy (3)
- Nanotechnology (10)
- National Security (2)
- Neutron Science (99)
- Physics (9)
- Quantum Computing (1)
- Quantum Science (7)
- Security (2)
- Space Exploration (3)
- Sustainable Energy (2)
- Transportation (5)
Media Contacts
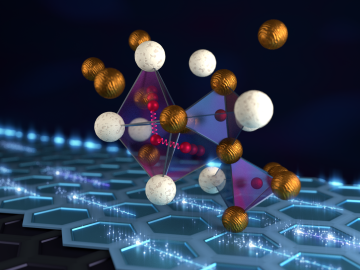
An international team of researchers has discovered the hydrogen atoms in a metal hydride material are much more tightly spaced than had been predicted for decades — a feature that could possibly facilitate superconductivity at or near room temperature and pressure.
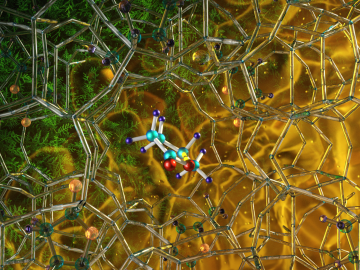
Illustration of the optimized zeolite catalyst, or NbAlS-1, which enables a highly efficient chemical reaction to create butene, a renewable source of energy, without expending high amounts of energy for the conversion. Credit: Jill Hemman, Oak Ridge National Laboratory/U.S. Dept. of Energy
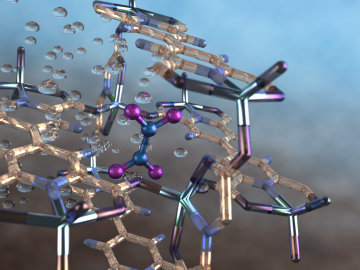
An international team of scientists, led by the University of Manchester, has developed a metal-organic framework, or MOF, material

Scientists at the U.S. Department of Energy’s Brookhaven National Laboratory have new experimental evidence and a predictive theory that solves a long-standing materials science mystery: why certain crystalline materials shrink when heated.
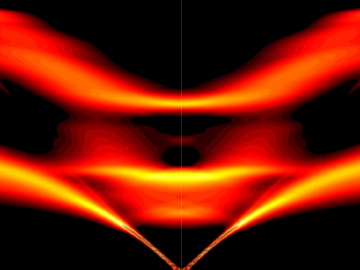
Scientists have discovered a way to alter heat transport in thermoelectric materials, a finding that may ultimately improve energy efficiency as the materials

Researchers at the Department of Energy’s Oak Ridge National Laboratory, Pacific Northwest National Laboratory and Washington State University teamed up to investigate the complex dynamics of low-water liquids that challenge nuclear waste processing at federal cleanup sites.
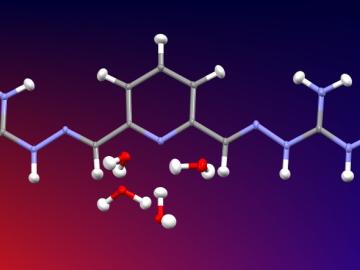
Researchers used neutron scattering at Oak Ridge National Laboratory’s Spallation Neutron Source to investigate the effectiveness of a novel crystallization method to capture carbon dioxide directly from the air.
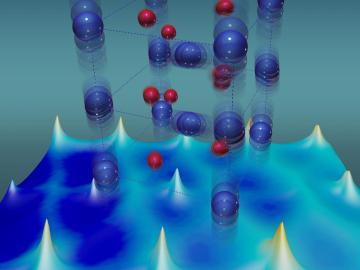
For more than 50 years, scientists have debated what turns particular oxide insulators, in which electrons barely move, into metals, in which electrons flow freely.




