Filter News
Area of Research
- (-) Materials (16)
- (-) Nuclear Science and Technology (4)
- Advanced Manufacturing (1)
- Biology and Environment (7)
- Clean Energy (50)
- Computational Engineering (2)
- Computer Science (5)
- Electricity and Smart Grid (1)
- Energy Sciences (1)
- Fusion and Fission (4)
- Fusion Energy (6)
- Isotopes (1)
- Materials for Computing (1)
- Mathematics (1)
- National Security (3)
- Neutron Science (3)
- Quantum information Science (1)
- Sensors and Controls (1)
- Supercomputing (5)
- Transportation Systems (2)
News Topics
- (-) Chemical Sciences (4)
- (-) Clean Water (1)
- (-) Energy Storage (7)
- (-) Fusion (3)
- (-) Molten Salt (4)
- (-) Transportation (6)
- 3-D Printing/Advanced Manufacturing (6)
- Advanced Reactors (5)
- Bioenergy (1)
- Biomedical (3)
- Buildings (1)
- Composites (4)
- Computer Science (1)
- Coronavirus (1)
- Critical Materials (5)
- Decarbonization (1)
- Environment (1)
- Isotopes (3)
- Materials (12)
- Materials Science (19)
- Microscopy (6)
- Nanotechnology (8)
- Neutron Science (6)
- Nuclear Energy (13)
- Physics (2)
- Polymers (6)
- Quantum Computing (1)
- Quantum Science (1)
- Space Exploration (3)
- Sustainable Energy (3)
Media Contacts

Electric vehicles can drive longer distances if their lithium-ion batteries deliver more energy in a lighter package. A prime weight-loss candidate is the current collector, a component that often adds 10% to the weight of a battery cell without contributing energy.

ORNL scientists found that a small tweak created big performance improvements in a type of solid-state battery, a technology considered vital to broader electric vehicle adoption.
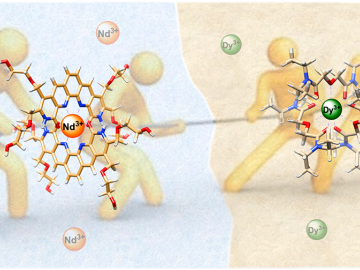
ORNL scientists combined two ligands, or metal-binding molecules, to target light and heavy lanthanides simultaneously for exceptionally efficient separation.
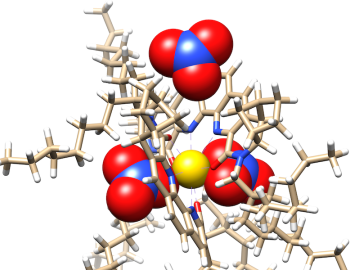
Researchers at ORNL zoomed in on molecules designed to recover critical materials via liquid-liquid extraction — a method used by industry to separate chemically similar elements.
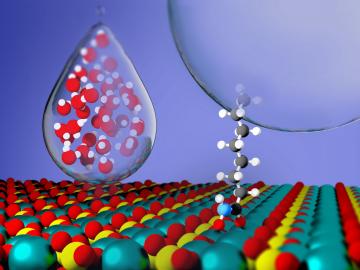
Critical Materials Institute researchers at Oak Ridge National Laboratory and Arizona State University studied the mineral monazite, an important source of rare-earth elements, to enhance methods of recovering critical materials for energy, defense and manufacturing applications.
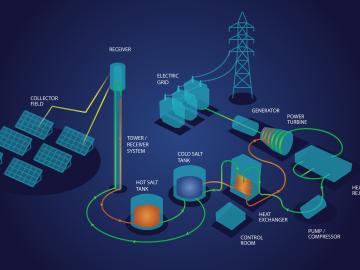
Oak Ridge National Laboratory scientists recently demonstrated a low-temperature, safe route to purifying molten chloride salts that minimizes their ability to corrode metals. This method could make the salts useful for storing energy generated from the sun’s heat.
Researchers at Oak Ridge National Laboratory are using state-of-the-art methods to shed light on chemical separations needed to recover rare-earth elements and secure critical materials for clean energy technologies.
Oak Ridge National Laboratory scientists seeking the source of charge loss in lithium-ion batteries demonstrated that coupling a thin-film cathode with a solid electrolyte is a rapid way to determine the root cause.

Oak Ridge National Laboratory researchers have developed a thin film, highly conductive solid-state electrolyte made of a polymer and ceramic-based composite for lithium metal batteries.

In the 1960s, Oak Ridge National Laboratory's four-year Molten Salt Reactor Experiment tested the viability of liquid fuel reactors for commercial power generation. Results from that historic experiment recently became the basis for the first-ever molten salt reactor benchmark.




