Filter News
Area of Research
- (-) Materials (13)
- (-) Materials for Computing (3)
- Advanced Manufacturing (1)
- Biology and Environment (14)
- Building Technologies (1)
- Clean Energy (40)
- Climate and Environmental Systems (2)
- Computational Biology (1)
- Computational Engineering (2)
- Computer Science (10)
- Fusion and Fission (1)
- Fusion Energy (8)
- Isotopes (1)
- Mathematics (1)
- National Security (3)
- Neutron Science (4)
- Nuclear Science and Technology (13)
- Nuclear Systems Modeling, Simulation and Validation (2)
- Quantum information Science (3)
- Supercomputing (19)
- Transportation Systems (2)
News Topics
- (-) Advanced Reactors (1)
- (-) Biomedical (3)
- (-) Computer Science (2)
- (-) Molten Salt (1)
- (-) Nuclear Energy (3)
- (-) Transportation (7)
- 3-D Printing/Advanced Manufacturing (8)
- Bioenergy (1)
- Buildings (1)
- Chemical Sciences (5)
- Clean Water (1)
- Composites (4)
- Coronavirus (3)
- Critical Materials (5)
- Decarbonization (1)
- Energy Storage (7)
- Environment (1)
- Fusion (2)
- Isotopes (2)
- Materials (18)
- Materials Science (25)
- Microscopy (8)
- Nanotechnology (11)
- Neutron Science (5)
- Physics (2)
- Polymers (7)
- Quantum Computing (1)
- Quantum Science (2)
- Space Exploration (1)
- Sustainable Energy (5)
Media Contacts

Electric vehicles can drive longer distances if their lithium-ion batteries deliver more energy in a lighter package. A prime weight-loss candidate is the current collector, a component that often adds 10% to the weight of a battery cell without contributing energy.

ORNL scientists found that a small tweak created big performance improvements in a type of solid-state battery, a technology considered vital to broader electric vehicle adoption.
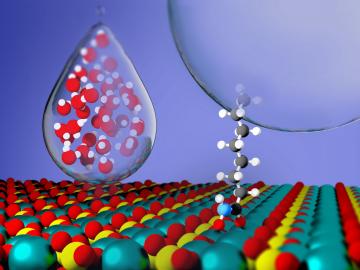
Critical Materials Institute researchers at Oak Ridge National Laboratory and Arizona State University studied the mineral monazite, an important source of rare-earth elements, to enhance methods of recovering critical materials for energy, defense and manufacturing applications.
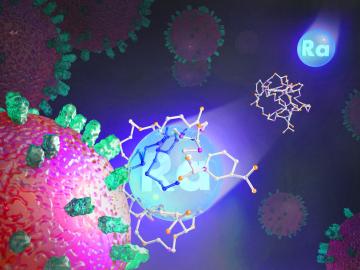
Researchers at ORNL explored radium’s chemistry to advance cancer treatments using ionizing radiation.

A discovery by Oak Ridge National Laboratory researchers may aid the design of materials that better manage heat.

Oak Ridge National Laboratory researchers have developed a new catalyst for converting ethanol into C3+ olefins – the chemical
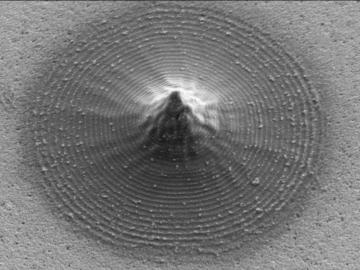
Scientists at Oak Ridge National Laboratory and the University of Tennessee designed and demonstrated a method to make carbon-based materials that can be used as electrodes compatible with a specific semiconductor circuitry.

Oak Ridge National Laboratory researchers have developed a thin film, highly conductive solid-state electrolyte made of a polymer and ceramic-based composite for lithium metal batteries.

Researchers at Oak Ridge National Laboratory proved that a certain class of ionic liquids, when mixed with commercially available oils, can make gears run more efficiently with less noise and better durability.

Using additive manufacturing, scientists experimenting with tungsten at Oak Ridge National Laboratory hope to unlock new potential of the high-performance heat-transferring material used to protect components from the plasma inside a fusion reactor. Fusion requires hydrogen isotopes to reach millions of degrees.




