Filter News
Area of Research
- (-) Energy Sciences (1)
- (-) Neutron Science (2)
- Advanced Manufacturing (1)
- Biology and Environment (6)
- Clean Energy (35)
- Computational Biology (1)
- Computational Engineering (1)
- Computer Science (5)
- Electricity and Smart Grid (1)
- Fusion and Fission (3)
- Fusion Energy (6)
- Isotopes (1)
- Materials (10)
- National Security (3)
- Nuclear Science and Technology (1)
- Quantum information Science (1)
- Sensors and Controls (1)
- Supercomputing (7)
- Transportation Systems (1)
News Topics
- (-) Energy Storage (3)
- Artificial Intelligence (1)
- Bioenergy (1)
- Biomedical (2)
- Chemical Sciences (1)
- Environment (1)
- Materials (3)
- Materials Science (3)
- Microscopy (1)
- Nanotechnology (1)
- Neutron Science (23)
- Nuclear Energy (1)
- Physics (1)
- Quantum Science (1)
- Space Exploration (1)
- Sustainable Energy (1)
- Transportation (1)
Media Contacts
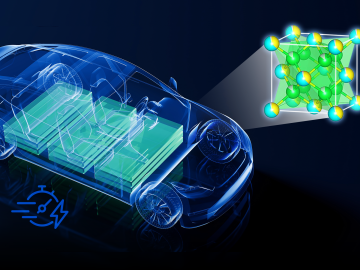
Currently, the biggest hurdle for electric vehicles, or EVs, is the development of advanced battery technology to extend driving range, safety and reliability.
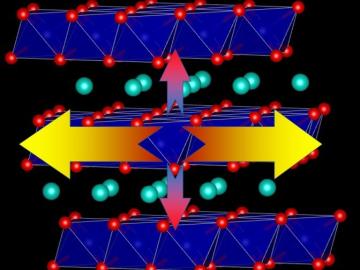
Oak Ridge National Laboratory researchers proved that the heat transport ability of lithium-ion battery cathodes is much lower than previously determined, a finding that could help explain barriers to increasing energy storage capacity and boosting performance.
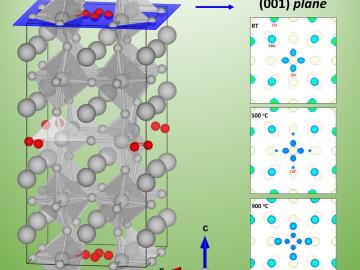
A University of South Carolina research team is investigating the oxygen reduction performance of energy conversion materials called perovskites by using neutron diffraction at Oak Ridge National Laboratory’s Spallation Neutron Source.




