Filter News
Area of Research
- Advanced Manufacturing (4)
- Biology and Environment (9)
- Building Technologies (2)
- Clean Energy (46)
- Computer Science (3)
- Electricity and Smart Grid (1)
- Energy Sciences (2)
- Fusion and Fission (4)
- Fusion Energy (6)
- Isotopes (1)
- Materials (15)
- Materials for Computing (2)
- National Security (2)
- Neutron Science (3)
- Nuclear Science and Technology (1)
- Quantum information Science (1)
- Sensors and Controls (1)
- Supercomputing (3)
- Transportation Systems (1)
News Type
News Topics
- (-) Chemical Sciences (9)
- (-) Decarbonization (8)
- (-) Energy Storage (31)
- (-) Fusion (9)
- (-) Grid (20)
- (-) Sustainable Energy (44)
- 3-D Printing/Advanced Manufacturing (31)
- Advanced Reactors (13)
- Artificial Intelligence (13)
- Big Data (16)
- Bioenergy (15)
- Biology (17)
- Biomedical (11)
- Biotechnology (3)
- Buildings (19)
- Clean Water (13)
- Climate Change (22)
- Composites (9)
- Computer Science (39)
- Coronavirus (11)
- Critical Materials (12)
- Cybersecurity (3)
- Environment (43)
- Exascale Computing (1)
- Frontier (1)
- High-Performance Computing (11)
- Hydropower (6)
- Irradiation (2)
- Isotopes (5)
- ITER (3)
- Machine Learning (10)
- Materials (35)
- Materials Science (33)
- Mathematics (1)
- Mercury (3)
- Microscopy (11)
- Molten Salt (5)
- Nanotechnology (12)
- National Security (3)
- Net Zero (1)
- Neutron Science (27)
- Nuclear Energy (19)
- Partnerships (1)
- Physics (4)
- Polymers (9)
- Quantum Computing (4)
- Quantum Science (10)
- Security (1)
- Simulation (7)
- Space Exploration (10)
- Statistics (1)
- Summit (6)
- Transportation (35)
Media Contacts
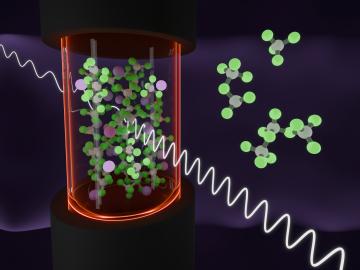
An Oak Ridge National Laboratory team revealed how chemical species form in a highly reactive molten salt mixture of aluminum chloride and potassium chloride by unraveling vibrational signatures and observing ion exchanges.
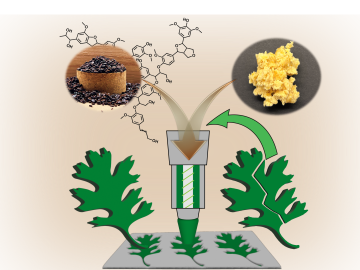
Oak Ridge National Laboratory scientists ingeniously created a sustainable, soft material by combining rubber with woody reinforcements and incorporating “smart” linkages between the components that unlock on demand.
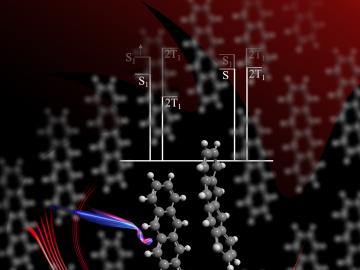
ORNL scientists develop a sample holder that tumbles powdered photochemical materials within a neutron beamline — exposing more of the material to light for increased photo-activation and better photochemistry data capture.

ORNL researchers used electron-beam additive manufacturing to 3D-print the first complex, defect-free tungsten parts with complex geometries.

A technology developed by Oak Ridge National Laboratory works to keep food refrigerated with phase change materials, or PCMs, while reducing carbon emissions by 30%.

An international team using neutrons set the first benchmark (one nanosecond) for a polymer-electrolyte and lithium-salt mixture. Findings could produce safer, more powerful lithium batteries.

ORNL researchers have developed a novel way to encapsulate salt hydrate phase-change materials within polymer fibers through a coaxial pulling process. The discovery could lead to the widespread use of the low-carbon materials as a source of insulation for a building’s envelope.

Electric vehicles can drive longer distances if their lithium-ion batteries deliver more energy in a lighter package. A prime weight-loss candidate is the current collector, a component that often adds 10% to the weight of a battery cell without contributing energy.
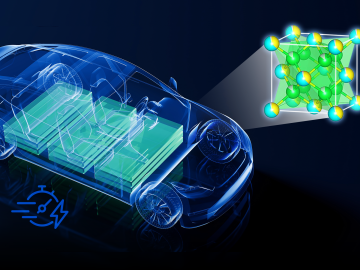
Currently, the biggest hurdle for electric vehicles, or EVs, is the development of advanced battery technology to extend driving range, safety and reliability.

Oak Ridge National Laboratory researchers are taking fast charging for electric vehicles, or EVs, to new extremes. A team of battery scientists recently developed a lithium-ion battery material that not only recharges 80% of its capacity in 10




