Filter News
Area of Research
- (-) Materials (4)
- Biology and Environment (3)
- Building Technologies (1)
- Clean Energy (10)
- Climate and Environmental Systems (1)
- Computational Engineering (2)
- Computer Science (10)
- Fusion Energy (2)
- Isotopes (3)
- Materials for Computing (1)
- Mathematics (1)
- National Security (2)
- Nuclear Science and Technology (4)
- Quantum information Science (3)
- Supercomputing (16)
News Topics
- (-) Computer Science (1)
- (-) Isotopes (2)
- (-) Molten Salt (1)
- 3-D Printing/Advanced Manufacturing (6)
- Advanced Reactors (1)
- Bioenergy (1)
- Biomedical (2)
- Buildings (1)
- Chemical Sciences (4)
- Clean Water (1)
- Composites (4)
- Coronavirus (1)
- Critical Materials (5)
- Decarbonization (1)
- Energy Storage (7)
- Environment (1)
- Fusion (2)
- Materials (12)
- Materials Science (19)
- Microscopy (6)
- Nanotechnology (8)
- Neutron Science (4)
- Nuclear Energy (3)
- Physics (2)
- Polymers (6)
- Quantum Computing (1)
- Quantum Science (1)
- Space Exploration (1)
- Sustainable Energy (3)
- Transportation (6)
Media Contacts
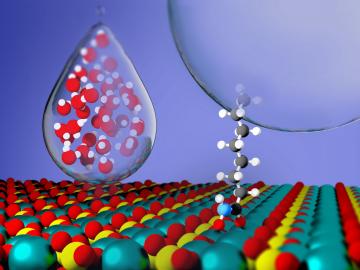
Critical Materials Institute researchers at Oak Ridge National Laboratory and Arizona State University studied the mineral monazite, an important source of rare-earth elements, to enhance methods of recovering critical materials for energy, defense and manufacturing applications.
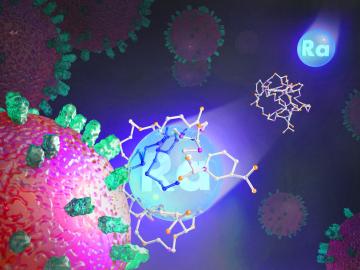
Researchers at ORNL explored radium’s chemistry to advance cancer treatments using ionizing radiation.

Oak Ridge National Laboratory scientists analyzed more than 50 years of data showing puzzlingly inconsistent trends about corrosion of structural alloys in molten salts and found one factor mattered most—salt purity.
Physicists turned to the “doubly magic” tin isotope Sn-132, colliding it with a target at Oak Ridge National Laboratory to assess its properties as it lost a neutron to become Sn-131.




