Filter News
Area of Research
- (-) Fusion and Fission (1)
- (-) Neutron Science (4)
- Advanced Manufacturing (1)
- Biology and Environment (2)
- Clean Energy (32)
- Computer Science (2)
- Electricity and Smart Grid (1)
- Energy Sciences (1)
- Isotopes (2)
- Materials (13)
- Materials for Computing (2)
- National Security (3)
- Nuclear Science and Technology (2)
- Quantum information Science (1)
- Sensors and Controls (1)
- Supercomputing (2)
News Topics
- (-) Energy Storage (3)
- (-) Microscopy (1)
- (-) Space Exploration (1)
- Advanced Reactors (1)
- Artificial Intelligence (1)
- Bioenergy (1)
- Biomedical (2)
- Chemical Sciences (2)
- Critical Materials (1)
- Environment (1)
- Fusion (2)
- ITER (2)
- Materials (3)
- Materials Science (3)
- Nanotechnology (1)
- Neutron Science (23)
- Nuclear Energy (1)
- Physics (1)
- Quantum Science (1)
- Transportation (1)
Media Contacts
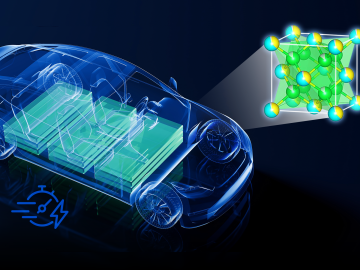
Currently, the biggest hurdle for electric vehicles, or EVs, is the development of advanced battery technology to extend driving range, safety and reliability.
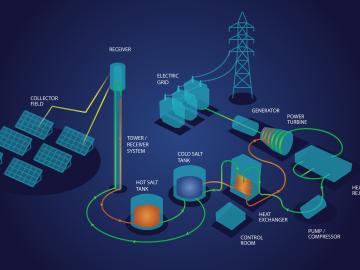
Oak Ridge National Laboratory scientists recently demonstrated a low-temperature, safe route to purifying molten chloride salts that minimizes their ability to corrode metals. This method could make the salts useful for storing energy generated from the sun’s heat.

Researchers from NASA’s Jet Propulsion Laboratory and Oak Ridge National Laboratory successfully created amorphous ice, similar to ice in interstellar space and on icy worlds in our solar system. They documented that its disordered atomic behavior is unlike any ice on Earth.
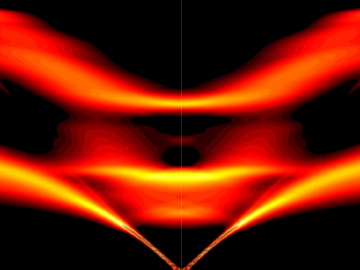
Scientists have discovered a way to alter heat transport in thermoelectric materials, a finding that may ultimately improve energy efficiency as the materials
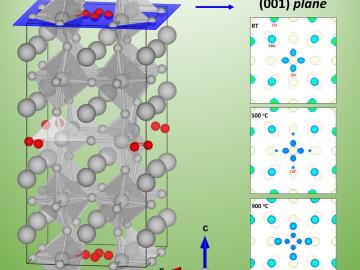
A University of South Carolina research team is investigating the oxygen reduction performance of energy conversion materials called perovskites by using neutron diffraction at Oak Ridge National Laboratory’s Spallation Neutron Source.




